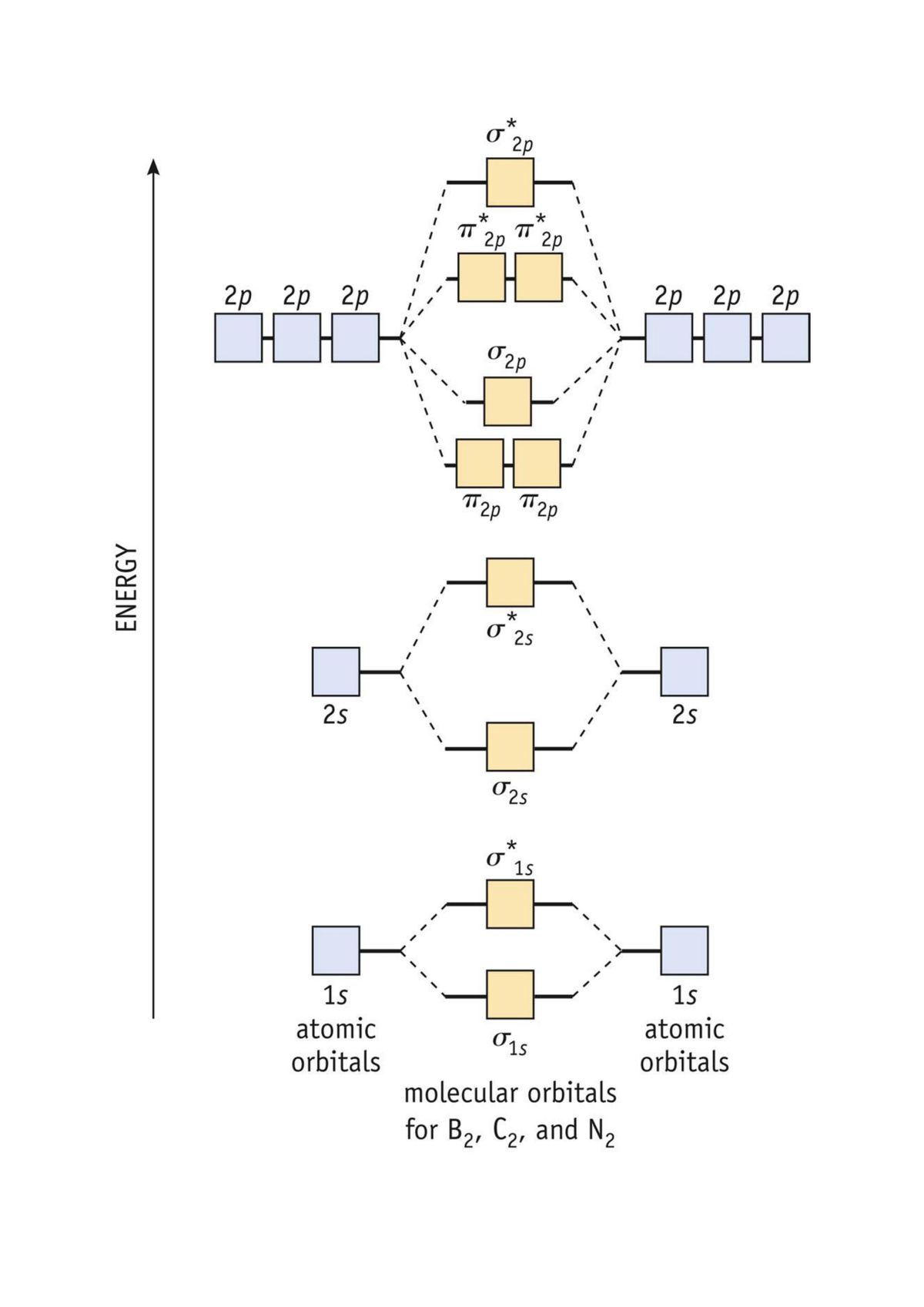
Understanding the intricacies of molecular orbitals is key to comprehend the behavior of molecules and their interactions. One of the most exemplifying tools in this regard is the C2 Molecular Orbital Diagram. This diagram provides a visual representation of the molecular orbitals spring when two carbon atoms combine to form a diatomic molecule. By study the C2 Molecular Orbital Diagram, we can gain insights into the tie and antibonding interactions that determine the molecule's constancy and reactivity.
Introduction to Molecular Orbitals
Molecular orbitals are mathematical functions that account the wave like deportment of electrons in molecules. They are formed by the combination of nuclear orbitals from individual atoms. When two atoms approach each other, their atomic orbitals overlap, leading to the constitution of molecular orbitals. These molecular orbitals can be adhere, antibonding, or nonbonding, depending on the phase and energy of the overlapping atomic orbitals.
The C2 Molecular Orbital Diagram
The C2 Molecular Orbital Diagram specifically focuses on the molecular orbitals formed by the combination of nuclear orbitals from two carbon atoms. Carbon has an electronic contour of 1s 2 2s2 2p2, which means it has four valence electrons. When two carbon atoms come together, their nuclear orbitals interact to form molecular orbitals.
To construct the C2 Molecular Orbital Diagram, we take to view the following steps:
- Identify the atomic orbitals of the case-by-case carbon atoms.
- Determine the possible combinations of these nuclear orbitals to form molecular orbitals.
- Arrange the molecular orbitals in order of increase energy.
- Fill the molecular orbitals with electrons according to the Aufbau principle, Hund's rule, and the Pauli excommunication principle.
Constructing the C2 Molecular Orbital Diagram
The first step in constructing the C2 Molecular Orbital Diagram is to identify the atomic orbitals of the individual carbon atoms. Each carbon atom has the follow nuclear orbitals:
- 1s orbital
- 2s orbital
- 2p orbitals (three degenerate orbitals: 2p x, 2p y, and 2p z )
When two carbon atoms approach each other, their nuclear orbitals overlap and form molecular orbitals. The 1s orbitals of the two carbon atoms combine to form a attach (σ1s) and an antibonding (σ 1s) molecular orbital. Similarly, the 2s orbitals combine to form a bind (σ2s) and an antibonding (σ 2s) molecular orbital. The 2p orbitals combine to form bind (π2p) and antibonding (π 2p) molecular orbitals, as well as a bonding (σ2p) and an antibonding (σ 2p) molecular orbital.
The molecular orbitals are arrange in order of increasing energy. The general order of energy levels for the C2 Molecular Orbital Diagram is as follows:
- σ1s
- σ 1s
- σ2s
- σ 2s
- π2p x
- π2p y
- σ2p z
- π 2p x
- π 2p y
- σ 2p z
Next, we fill the molecular orbitals with electrons according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle. Each molecular orbital can hold a maximum of two electrons with opposite spins. The electronic constellation of C2 is shape by placing the eight valence electrons (four from each carbon atom) into the molecular orbitals in order of increase energy.
The leave electronic configuration for C2 is:
- σ1s 2
- σ 1s 2
- σ2s 2
- σ 2s 2
- π2p x1
- π2p y1
This configuration indicates that the C2 molecule has two unpaired electrons in the π2p orbitals, making it paramagnetic.
Interpreting the C2 Molecular Orbital Diagram
The C2 Molecular Orbital Diagram provides valuable insights into the bind and constancy of the C2 molecule. The presence of unpaired electrons in the π2p orbitals suggests that the molecule is responsive and can readily form bonds with other atoms or molecules. Additionally, the diagram helps us realize the energy levels of the molecular orbitals, which is all-important for predicting the molecule s spectroscopic properties.
One significant aspect of the C2 Molecular Orbital Diagram is the concept of bond order. Bond order is a measure of the number of bonding electron pairs minus the turn of antibonding electron pairs. For the C2 molecule, the bond order can be calculated as follows:
- Number of stick electrons: 6 (2 in σ1s, 2 in σ2s, and 2 in π2p)
- Number of antibonding electrons: 4 (2 in σ 1s and 2 in σ 2s)
Bond order (Number of bonding electrons Number of antibonding electrons) 2
Bond order (6 4) 2 1
This indicates that the C2 molecule has a single bond between the two carbon atoms, which is consistent with its paramagnetic nature and reactivity.
Another significant concept is the energy difference between the highest fill molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). This energy gap, known as the HOMO LUMO gap, is important for realise the molecule's chemic reactivity and constancy. A smaller HOMO LUMO gap indicates higher reactivity, as it is easier for electrons to transition from the HOMO to the LUMO.
In the case of the C2 molecule, the HOMO is the π2p orbital, and the LUMO is the σ 2p orbital. The energy difference between these orbitals is comparatively pocket-size, which explains the eminent reactivity of the C2 molecule.
Applications of the C2 Molecular Orbital Diagram
The C2 Molecular Orbital Diagram has legion applications in chemistry and materials science. It is used to predict the properties of diatomic molecules, understand their reactivity, and design new materials with specific properties. for instance, the diagram can help in the development of carbon establish materials, such as graphene and carbon nanotubes, which have unequaled electronic and mechanical properties.
Additionally, the C2 Molecular Orbital Diagram is used in spectroscopical studies to interpret the assimilation and discharge spectra of molecules. By see the energy levels of the molecular orbitals, scientists can predict the wavelengths of light that a molecule will absorb or emit, providing valuable info about its construction and dynamics.
In the field of catalysis, the C2 Molecular Orbital Diagram is used to design catalysts that can help chemic reactions. By read the bonding and antibonding interactions in molecules, scientists can make catalysts that lower the activating energy of reactions, make them more effective and selective.
Comparing C2 with Other Diatomic Molecules
To gain a deeper understanding of the C2 Molecular Orbital Diagram, it is helpful to compare it with the molecular orbital diagrams of other diatomic molecules. for instance, view the molecular orbital diagrams of N2 and O2.
Nitrogen (N2) has an electronic shape of 1s 2 2s2 2p3, and oxygen (O2) has an electronic constellation of 1s 2 2s2 2p4. The molecular orbital diagrams of N2 and O2 are similar to that of C2, but with differences in the number of electrons and the energy levels of the molecular orbitals.
For N2, the molecular orbital diagram shows that all electrons are paired, lead in a bond order of 3. This eminent bond order explains the strong triple bond between the two nitrogen atoms, making N2 a very stable molecule.
For O2, the molecular orbital diagram shows two unpaired electrons in the π 2p orbitals, resulting in a bond order of 2. This indicates a double bond between the two oxygen atoms, and the presence of unpaired electrons makes O2 paramagnetic.
Comparing these diagrams with the C2 Molecular Orbital Diagram helps us realise the differences in tie, stability, and reactivity among these diatomic molecules.
Here is a comparison table for C2, N2, and O2:
| Molecule | Electronic Configuration | Bond Order | Magnetic Properties |
|---|---|---|---|
| C2 | σ1s 2 σ*1s2 σ2s2 σ*2s2 π2px1 π2py1 | 1 | Paramagnetic |
| N2 | σ1s 2 σ*1s2 σ2s2 σ*2s2 π2px2 π2py2 σ2pz2 | 3 | Diamagnetic |
| O2 | σ1s 2 σ*1s2 σ2s2 σ*2s2 σ2pz2 π2px2 π2py2 π*2px1 π*2py1 | 2 | Paramagnetic |
Note: The comparison table highlights the differences in electronic conformation, bond order, and magnetic properties among C2, N2, and O2. This comparison helps in understanding the alone characteristics of each molecule found on their molecular orbital diagrams.
Visualizing the C2 Molecular Orbital Diagram
Visualizing the C2 Molecular Orbital Diagram can greatly enhance our understand of the molecular orbitals and their interactions. While detail diagrams are typically found in textbooks or scientific lit, a basic representation can be drawn to exemplify the key points.
Below is a simplify representation of the C2 Molecular Orbital Diagram:

This diagram shows the energy levels of the molecular orbitals and the dispersion of electrons. The bonding orbitals are lower in energy and are filled first, followed by the antibonding orbitals. The unpaired electrons in the π2p orbitals are highlight, bespeak the paramagnetic nature of the C2 molecule.
By studying this diagram, we can gain a deeper understanding of the bonding and antibonding interactions in the C2 molecule, as easily as its electronic structure and reactivity.
to sum, the C2 Molecular Orbital Diagram is a powerful tool for understanding the electronic construction and bond in diatomic molecules. By examining the molecular orbitals and their interactions, we can predict the properties of molecules, design new materials, and gain insights into chemic reactivity. The diagram provides a visual representation of the energy levels and electron dispersion, aid us to comprehend the complex behavior of molecules at the atomic stage. This noesis is crucial for advancing our translate of chemistry and materials skill, and for developing innovative technologies that rely on molecular interactions.
Related Terms:
- electronic configuration of c2 molecule
- n2 mo diagram
- energy level diagram of c2
- orbital diagram of c2
- how does c2 bond
- molecular orbital theory for c2