Understanding the molecular geometry of a compound is essential for foreshadow its chemical behavior and properties. One such compound that has earn significant interest is ClF3, or chlorine trifluoride. This molecule's alone construction and properties make it a enamor subject for study. In this post, we will delve into the ClF3 molecular geometry, its bonding, and the factors that influence its shape.
Introduction to ClF3
Chlorine trifluoride (ClF3) is a highly responsive and toxic compound composed of one chlorine atom and three fluorine atoms. It is known for its potent oxidate properties and is often used in industrial applications, particularly in the production of uranium hexafluoride (UF6) for nuclear fuel processing. The molecule's reactivity and constancy are nearly tied to its molecular geometry, which we will explore in detail.
Understanding Molecular Geometry
Molecular geometry refers to the three dimensional arrangement of atoms in a molecule. This arrangement is set by the bit of electron pairs around the central atom and the horror between these pairs. The ClF3 molecular geometry can be understood using the Valence Shell Electron Pair Repulsion (VSEPR) theory, which predicts the shape of molecules found on the repugnance between electron pairs.
VSEPR Theory and ClF3
The VSEPR theory helps us predict the shape of the ClF3 molecule by considering the repulsion between electron pairs. Chlorine, the central atom, has seven valence electrons, and each fluorine atom contributes one electron to form a covalent bond. This results in three bonding pairs and two lone pairs around the chlorine atom.
According to VSEPR theory, the five electron pairs (three attach and two lone) will arrange themselves in a rhombohedral bipyramidal geometry to belittle repulsion. However, the presence of lone pairs affects the terminal shape. Lone pairs repel more strongly than bonding pairs, have the molecule to distort from the idealistic rhombohedral bipyramidal shape.
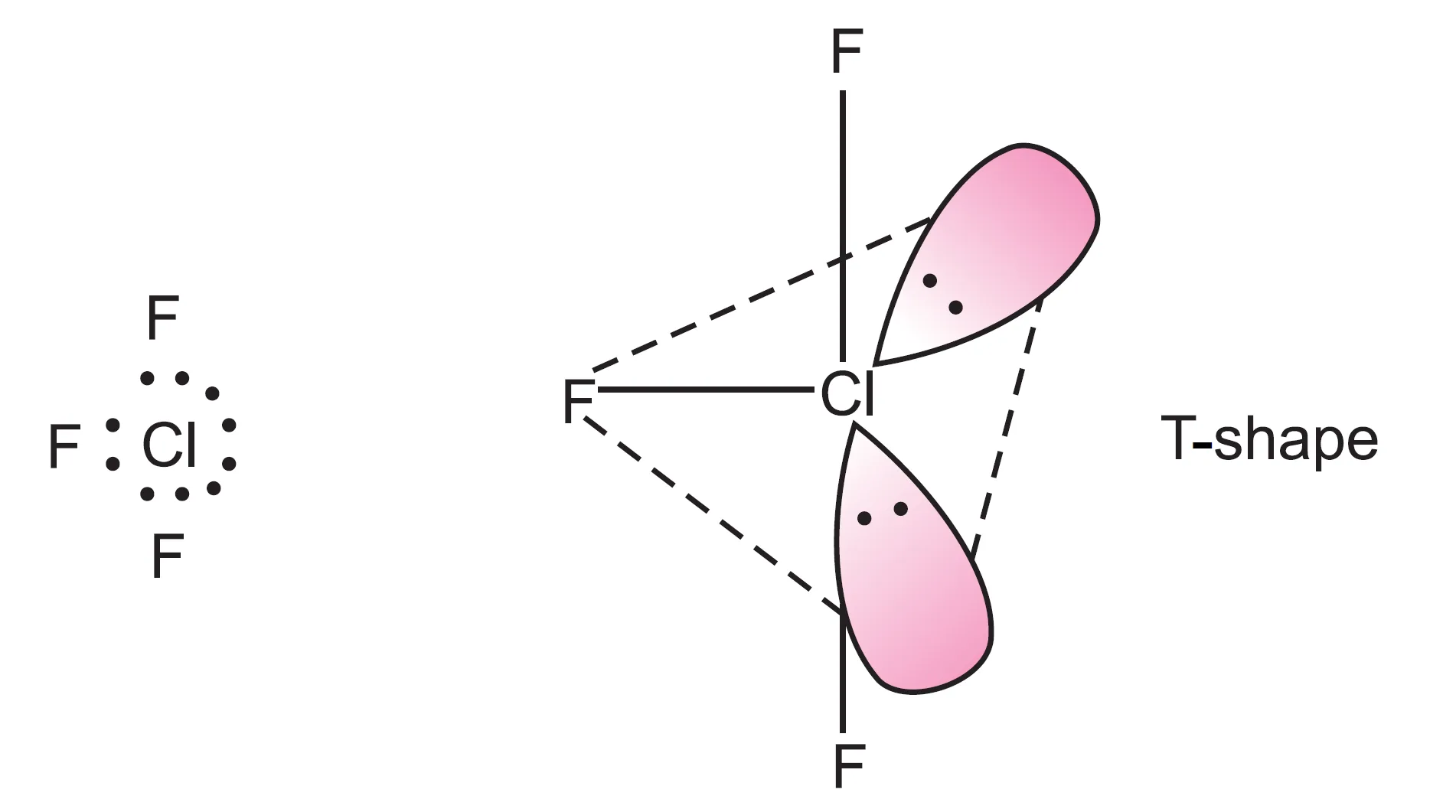
ClF3 Molecular Geometry: T Shaped Structure
The ClF3 molecular geometry is best report as T shaped. This deformation occurs because the two lone pairs on the chlorine atom repel the bind pairs more strongly, pushing the fluorine atoms finisher together. The result shape has the three fluorine atoms forming a T with the chlorine atom at the center.
To visualize this, imagine a rhombohedral bipyramidal arrangement where the two axile positions are occupied by lone pairs. The three equatorial positions are fill by the fluorine atoms, forming a T shape. This arrangement minimizes the repulsion between the electron pairs and results in the observed molecular geometry.
Bond Angles and Bond Lengths
The bond angles in ClF3 are not equal due to the presence of lone pairs. The F Cl F bond angles are about 87. 5 degrees, which is significantly less than the idealistic 120 degrees for a rhombohedral planar arrangement. This decrease is due to the repulsion from the lone pairs, which pushes the fluorine atoms finisher together.
The bond lengths in ClF3 are also mold by the electronic construction. The Cl F bond length is typically around 1. 63 Å, which is shorter than ask due to the strong negativity of fluorine. The strong attraction between the chlorine and fluorine atoms results in a shorter bond length, contributing to the molecule's constancy.
Electronic Structure and Hybridization
The electronic construction of ClF3 involves the hybridization of the chlorine atom's orbitals. Chlorine uses sp3d hybridization to form bonds with the fluorine atoms. This hybridization allows the chlorine atom to accommodate the five electron pairs (three bonding and two lone) in a trigonal bipyramidal arrangement.
The sp3d crossing involves the meld of one s orbital, three p orbitals, and one d orbital from the chlorine atom. This results in five sp3d hybrid orbitals, which can adapt the five electron pairs. The three tie pairs form sigma bonds with the fluorine atoms, while the two lone pairs occupy the remaining hybrid orbitals.
Properties of ClF3
ClF3 is a highly responsive and toxic compound with several celebrated properties:
- Oxidizing Agent: ClF3 is a strong oxidizing agent and can react violently with many substances, include h2o, organic materials, and metals.
- Toxicity: The compound is exceedingly toxic and can cause severe burns and respiratory issues upon exposure.
- Stability: Despite its reactivity, ClF3 is relatively stable under controlled conditions and is used in industrial processes.
- Melting and Boiling Points: ClF3 has a melting point of 76. 3 C and a boiling point of 11. 8 C, designate its volatile nature.
Applications of ClF3
ClF3 has several industrial applications due to its potent oxidate properties:
- Nuclear Fuel Processing: ClF3 is used in the product of uranium hexafluoride (UF6), which is essential for the enrichment of uranium in nuclear fuel.
- Rocket Propellant: The compound has been regard as a likely rocket propelling due to its high energy content and reactivity.
- Chemical Synthesis: ClF3 is used in various chemic synthesis processes, particularly in the production of fluorinated compounds.
Safety Considerations
Handling ClF3 requires extreme caveat due to its high reactivity and toxicity. Safety measures include:
- Personal Protective Equipment (PPE): Use allow PPE, including gloves, goggles, and protective clothing, to minimise exposure.
- Ventilation: Ensure proper airing to prevent the accretion of toxic fumes.
- Storage: Store ClF3 in a cool, dry position away from antagonistic substances.
- Emergency Procedures: Have emergency procedures in place for manage spills and exposures, include access to aesculapian treatment.
Note: Always follow safety guidelines and regulations when handling hazardous chemicals like ClF3.
Conclusion
The ClF3 molecular geometry is a fascinating example of how electronic structure and repulsion between electron pairs influence the shape of a molecule. The T shaped structure of ClF3, determined by VSEPR theory, highlights the importance of lone pairs in molecular geometry. Understanding the bonding, hybridizing, and properties of ClF3 provides valuable insights into its reactivity and applications. Whether in nuclear fuel processing or chemical synthesis, ClF3 s unique characteristics make it a crucial compound in diverse industrial processes. However, its care requires extreme precaution due to its eminent reactivity and toxicity. By appreciating the molecular geometry and properties of ClF3, we can punter realize its role in chemistry and industry.
Related Terms:
- clf3 polar or non polar
- clf5 molecular geometry
- clf3 shape
- clf3 bond angle
- clf3 lewis
- clf3 electron geometry