Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful analytic technique used to find the structure and dynamics of molecules. One of the underlying concepts in NMR is the distinction between downfield vs upfield NMR. Understanding this preeminence is important for interpreting NMR spectra and gaining insights into molecular structures.
Understanding NMR Spectroscopy
NMR spectroscopy relies on the magnetised properties of certain atomic nuclei. When placed in a magnetized field, these nuclei absorb and re emit electromagnetic radiation at specific frequencies. The resulting spectrum provides info about the chemic environment of the nuclei, which can be used to identify functional groups and molecular structures.
Chemical Shift in NMR
The chemical shift is a key parameter in NMR spectroscopy. It measures the plangency frequency of a nucleus relative to a standard acknowledgment. Chemical shifts are expressed in parts per million (ppm) and are influenced by the electronic environment around the nucleus. The chemic shift scale is divide into two main regions: downfield and upfield.
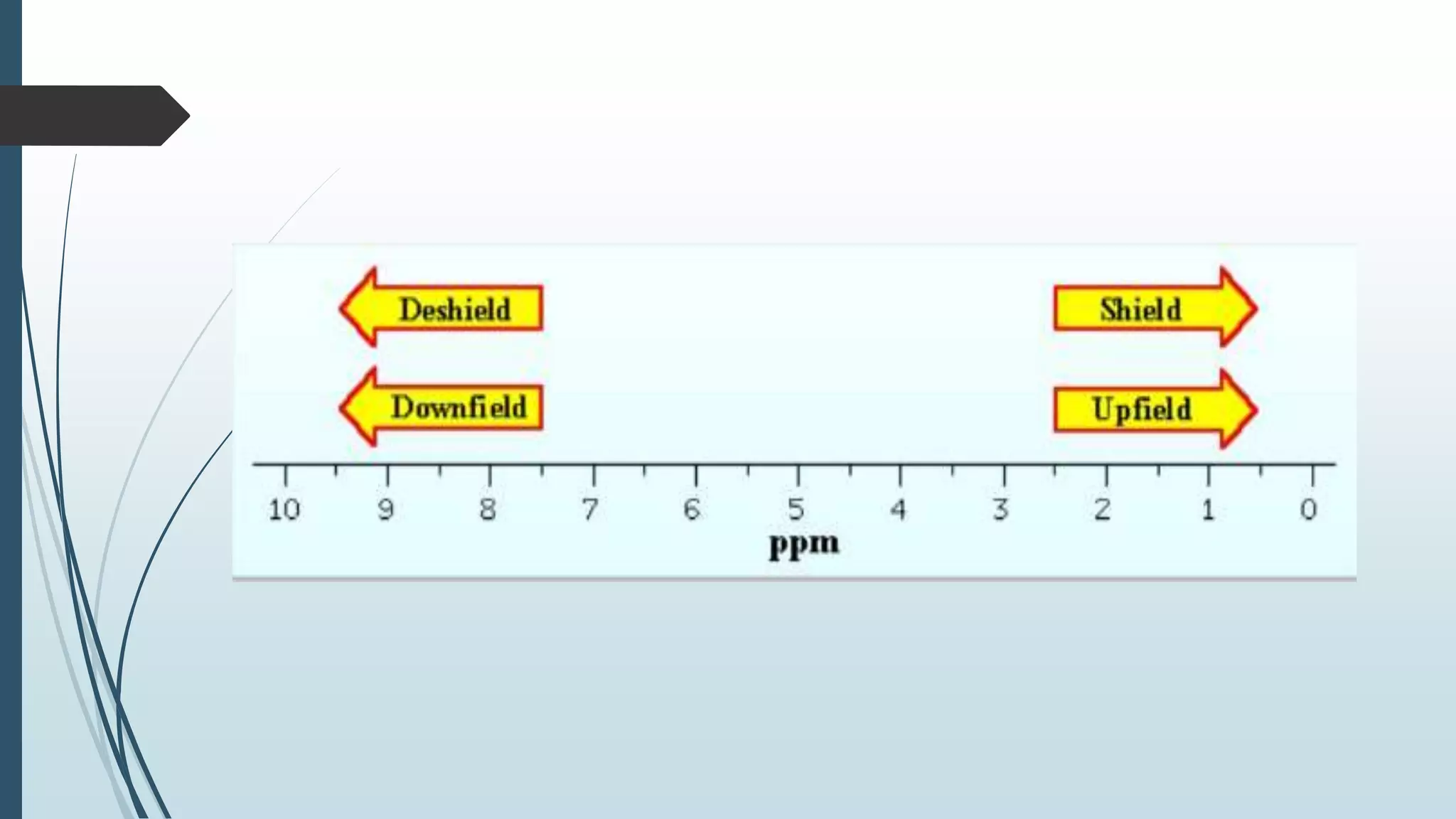
Downfield vs Upfield NMR
Downfield and upfield are terms used to describe the perspective of signals in an NMR spectrum proportional to a reference standard, typically tetramethylsilane (TMS).
Downfield NMR
Downfield refers to the region of the NMR spectrum where signals appear at higher ppm values. Nuclei that are deshielded, entail they experience a reduce electron density, resonate at higher frequencies and appear downfield. Common examples of downfield signals include:
- Protons attach to electronegative atoms (e. g., oxygen, nitrogen, halogens).
- Protons in redolent compounds.
- Protons in carbonyl groups.
Downfield shifts are frequently observed in functional groups that have electron disengage substituents, which reduce the electron concentration around the nucleus. This deshielding effect causes the nucleus to resonate at a higher frequency, ensue in a downfield shift.
Upfield NMR
Upfield refers to the region of the NMR spectrum where signals appear at lower ppm values. Nuclei that are screen, mean they experience an increased electron density, resonate at lower frequencies and appear upfield. Common examples of upfield signals include:
- Protons in alkyl groups (e. g., methyl, ethyl).
- Protons in saturate hydrocarbons.
- Protons in molecules with electron donating substituents.
Upfield shifts are often observed in functional groups that have electron donating substituents, which increase the electron concentration around the nucleus. This shielding effect causes the nucleus to vibrate at a lower frequency, resulting in an upfield shift.
Factors Affecting Chemical Shift
Several factors influence the chemical shift of a nucleus in an NMR spectrum. Understanding these factors is crucial for interpreting downfield vs upfield NMR signals accurately.
Electronegativity
Electronegative atoms withdraw electron density from neighbor atoms, causing a deshielding effect and a downfield shift. for example, protons attached to oxygen or nitrogen atoms in alcohols, amines, and ethers typically vibrate downfield.
Inductive Effects
Inductive effects occur when electron withdraw or electron donating groups influence the electron concentration of neighboring atoms. Electron retire groups (e. g., halogens, carbonyl groups) cause a downfield shift, while electron donating groups (e. g., alkyl groups) cause an upfield shift.
Anisotropic Effects
Anisotropic effects arise from the magnetised anisotropy of certain functional groups, such as aromatic rings and carbonyl groups. These groups create local magnetized fields that can either shield or deshield nearby nuclei, leading to upfield or downfield shifts, respectively.
Hydrogen Bonding
Hydrogen bonding can also touch the chemic shift of protons. When a proton is involved in hydrogen bonding, it experiences a deshielding effect, result in a downfield shift. This is commonly observed in alcohols and carboxylic acids.
Interpreting NMR Spectra
Interpreting NMR spectra involves identifying the chemic shifts of different protons and correlate them with the molecular structure. Here are some steps to help you interpret downfield vs upfield NMR signals:
Identify the Reference Standard
The reference standard, typically TMS, is set at 0 ppm. All other signals are measure comparative to this standard.
Analyze the Chemical Shifts
Determine whether the signals are downfield or upfield by comparing their ppm values to the reference standard. Downfield signals have higher ppm values, while upfield signals have lower ppm values.
Correlate with Functional Groups
Use the chemic shift values to identify the functional groups exhibit in the molecule. Refer to standard chemical shift tables to match the detect signals with known functional groups.
Consider Integration and Splitting Patterns
Integration provides info about the bit of protons responsible for each signal, while break patterns (e. g., singlet, doublet, triplet) bespeak the turn of neighboring protons. These factors, along with chemic shifts, assist in allot the signals to specific protons in the molecule.
Note: Always take the overall molecular structure and the chemical environment of each proton when see NMR spectra.
Applications of NMR Spectroscopy
NMR spectroscopy has a blanket range of applications in respective fields, include chemistry, biology, and materials skill. Some key applications include:
Structural Elucidation
NMR spectroscopy is extensively used to ascertain the construction of organic compounds. By analyzing the chemic shifts, integration, and divide patterns, chemists can deduce the molecular construction, include the arrangement of atoms and functional groups.
Conformational Analysis
NMR spectroscopy provides insights into the conformational dynamics of molecules. By studying the chemical shifts and coupling constants, researchers can understand the preferred conformations and the energy barriers between different conformations.
Biomolecular Studies
NMR spectroscopy is a valuable tool for studying biomolecules, such as proteins and nucleic acids. It helps in determining the three dimensional structure, dynamics, and interactions of biomolecules, which are important for understanding their biological functions.
Materials Science
In materials science, NMR spectroscopy is used to characterize the structure and dynamics of polymers, ceramics, and other materials. It provides information about the molecular organization, mobility, and interactions within these materials.
Advanced NMR Techniques
besides one dimensional (1D) NMR spectroscopy, various advanced techniques are uncommitted for more detailed structural analysis. These techniques furnish additional dimensions of information, heighten the declaration and sensitivity of NMR spectra.
Two Dimensional (2D) NMR
2D NMR techniques, such as COSY (Correlation Spectroscopy), NOESY (Nuclear Overhauser Effect Spectroscopy), and HSQC (Heteronuclear Single Quantum Coherence), furnish info about the connectivity and spacial propinquity of nuclei. These techniques are particularly useful for ascribe signals to specific protons and for determining the three dimensional construction of molecules.
Solid State NMR
Solid state NMR spectroscopy is used to study the structure and dynamics of solids. It provides info about the local environment of nuclei in crystalline and amorphous materials, create it a worthful puppet for materials skill and catalysis enquiry.
Dynamic Nuclear Polarization (DNP)
DNP is an supercharge technique that enhances the sensitivity of NMR spectroscopy by transferring polarization from unpaired electrons to nuclei. This technique is particularly utilitarian for analyse low abundance nuclei and for ameliorate the signal to noise ratio in NMR spectra.
Note: Advanced NMR techniques expect specialized equipment and expertise. Consult with an NMR specializer for optimal results.
Challenges and Limitations
While NMR spectroscopy is a potent analytical tool, it also has its challenges and limitations. Some of the key challenges include:
Sensitivity
NMR spectroscopy has relatively low sensitivity compared to other spectroscopic techniques, such as mass spectrometry and infrared spectroscopy. This limit can be overcome using advanced techniques like DNP and by increase the sample density.
Resolution
The resolution of NMR spectra can be touch by various factors, including magnetic battlefield homogeneity, sample honour, and molecular dynamics. High resolution NMR spectrometers and advanced pulse sequences can help better the resolution of NMR spectra.
Interpretation Complexity
Interpreting NMR spectra can be complex, especially for large molecules with many overlapping signals. Advanced NMR techniques and computational tools can aid in the version of complex spectra.
Future Directions
The field of NMR spectroscopy continues to evolve, with new techniques and applications being germinate. Some of the future directions in NMR spectroscopy include:
High Field NMR
High battlefield NMR spectrometers, run at magnetic fields of 1 GHz and above, offer better sensitivity and resolution. These spectrometers are expected to become more widely available, enable more detail structural studies.
In Vivo NMR
In vivo NMR spectroscopy is used to study biological processes in living organisms. This technique has applications in medical diagnostics, drug development, and metabolous studies.
Machine Learning and AI
Machine memorise and hokey intelligence are being increasingly used to analyze NMR spectra. These technologies can help in automating the rendition of spectra, identifying patterns, and predicting molecular structures.
Note: Stay update with the latest developments in NMR spectroscopy to leverage new techniques and applications.
to sum, see downfield vs upfield NMR is fundamental to interpreting NMR spectra and gaining insights into molecular structures. By canvas chemical shifts, integration, and splitting patterns, researchers can deduce the construction and dynamics of molecules. Advanced NMR techniques and hereafter developments continue to enhance the capabilities of NMR spectroscopy, make it an indispensable puppet in various scientific disciplines.
Related Terms:
- nmr downfield shift
- highest field signal nmr
- down battlefield in nmr
- upfield shift in nmr
- harbour and deshielded
- upfield and downfield in nmr