Understanding the intricacies of Electron And Molecular Geometry is fundamental to dig the demeanour of molecules and their interactions. This knowledge is all-important in fields such as chemistry, materials science, and biochemistry. By delving into the principles of electron and molecular geometry, we can predict the shapes of molecules, understand their reactivity, and design new materials with specific properties.
Understanding Electron Geometry
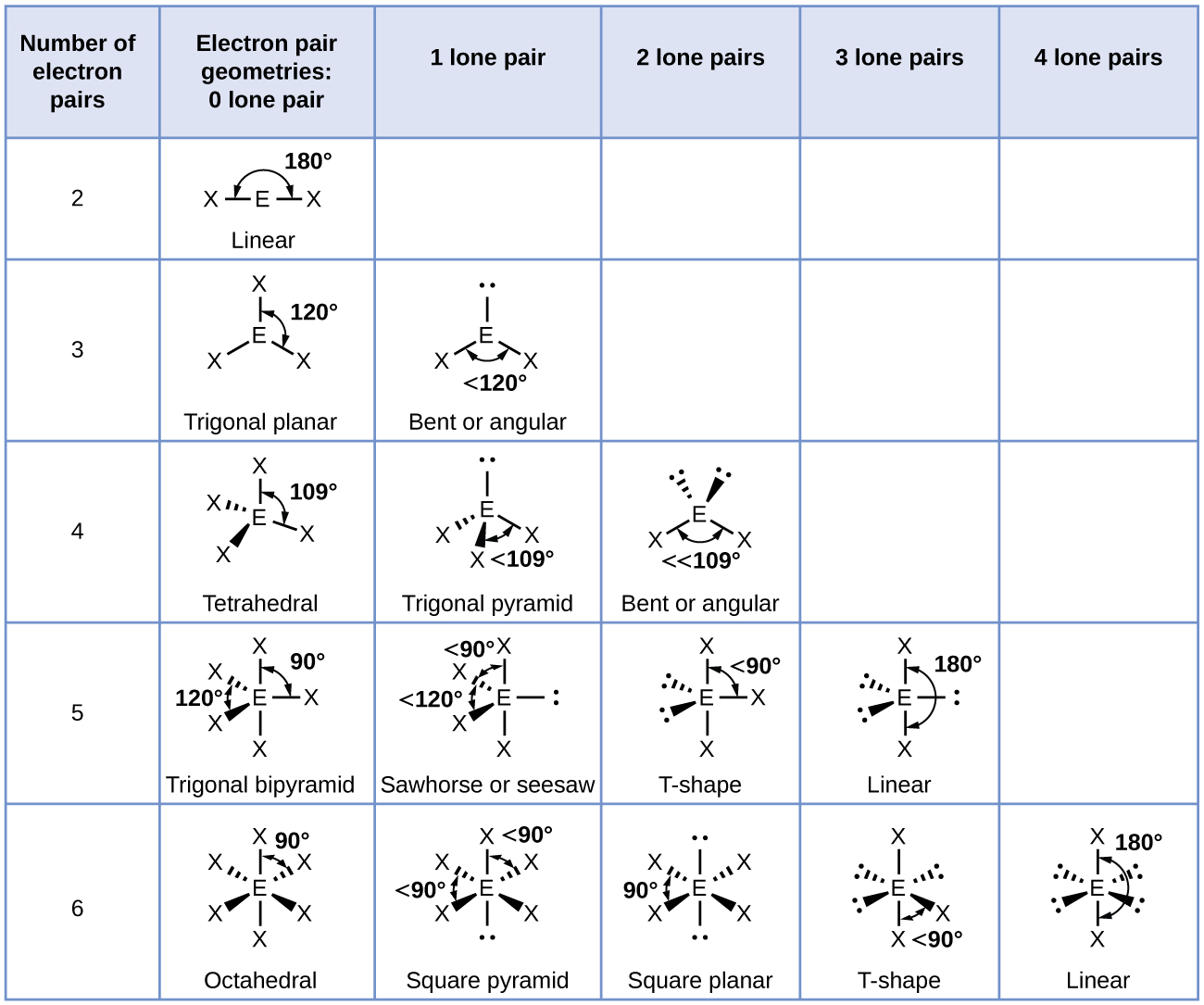
Electron geometry refers to the spatial arrangement of electron pairs around a primal atom in a molecule. This arrangement is ascertain by the repulsion between electron pairs, which search to minimize their common repugnance. The key concept here is the Valence Shell Electron Pair Repulsion (VSEPR) theory, which helps predict the geometry of molecules establish on the number of electron pairs around the cardinal atom.
VSEPR theory postulates that electron pairs around a central atom will repel each other and arrange themselves in a way that maximizes the distance between them. This results in specific geometries count on the figure of electron pairs:
- Two electron pairs: Linear geometry (e. g., BeCl2)
- Three electron pairs: Trigonal planar geometry (e. g., BF3)
- Four electron pairs: Tetrahedral geometry (e. g., CH4)
- Five electron pairs: Trigonal bipyramidal geometry (e. g., PF5)
- Six electron pairs: Octahedral geometry (e. g., SF6)
Molecular Geometry vs. Electron Geometry
While electron geometry considers all electron pairs (both tie and non bonding), molecular geometry focuses only on the positions of the atoms in the molecule. The difference between the two arises from the front of lone pairs, which occupy more space than bonding pairs due to their higher revulsion.
for representative, in the h2o molecule (H2O), the central oxygen atom has two tie pairs and two lone pairs. The electron geometry is tetrahedral, but the molecular geometry is bent due to the horror from the lone pairs.
Factors Affecting Electron And Molecular Geometry
Several factors influence the electron and molecular geometry of a molecule:
- Number of Electron Pairs: The more electron pairs around the key atom, the more complex the geometry.
- Presence of Lone Pairs: Lone pairs repel more powerfully than attach pairs, involve the overall shape.
- Electronegativity: The difference in negativity between atoms can influence the distribution of electron density.
- Steric Hindrance: Bulky groups can cause steric hindrance, affecting the spatial arrangement of atoms.
Common Molecular Geometries
Understanding the common molecular geometries is all-important for forecast the doings of molecules. Here are some of the most mutual geometries:
| Number of Bonding Pairs | Number of Lone Pairs | Electron Geometry | Molecular Geometry | Example |
|---|---|---|---|---|
| 2 | 0 | Linear | Linear | CO2 |
| 3 | 0 | Trigonal Planar | Trigonal Planar | BF3 |
| 4 | 0 | Tetrahedral | Tetrahedral | CH4 |
| 3 | 1 | Trigonal Planar | Bent | SO2 |
| 2 | 2 | Tetrahedral | Bent | H2O |
| 5 | 0 | Trigonal Bipyramidal | Trigonal Bipyramidal | PF5 |
| 4 | 1 | Trigonal Bipyramidal | Seesaw | SF4 |
| 6 | 0 | Octahedral | Octahedral | SF6 |
| 5 | 1 | Octahedral | Square Pyramidal | BrF5 |
Note: The presence of lone pairs can importantly alter the molecular geometry liken to the electron geometry. Always consider the number and place of lone pairs when shape the shape of a molecule.
Applications of Electron And Molecular Geometry
The study of Electron And Molecular Geometry has blanket roam applications in respective fields:
- Chemical Reactivity: Understanding the geometry of molecules helps predict their reactivity and the types of reactions they can undergo.
- Material Science: Designing new materials with specific properties, such as conduction or strength, relies on translate molecular geometry.
- Pharmaceuticals: The shape of molecules is all-important in drug design, as it affects how a drug interacts with its target.
- Environmental Science: Knowing the geometry of pollutants can aid in evolve strategies for their removal or abasement.
Predicting Molecular Geometry
Predicting the molecular geometry of a compound involves respective steps:
- Identify the cardinal atom and the surrounding atoms.
- Determine the number of adhere and non bonding electron pairs around the key atom.
- Use VSEPR theory to predict the electron geometry.
- Adjust for the presence of lone pairs to determine the molecular geometry.
for illustration, consider the molecule NH3 (ammonia):
- The cardinal atom is nitrogen (N).
- There are three bonding pairs (N H) and one lone pair on the nitrogen atom.
- The electron geometry is tetrahedral.
- The molecular geometry is trigonal pyramidal due to the lone pair.
Note: Always draw the Lewis structure of the molecule to accurately determine the bit of adhere and non bonding electron pairs.
Advanced Topics in Electron And Molecular Geometry
Beyond the basics, there are advanced topics that delve deeper into the complexities of Electron And Molecular Geometry:
- Hybridization: The commingle of nuclear orbitals to form hybrid orbitals, which can explain the bonding in molecules with complex geometries.
- Molecular Orbital Theory: A more advanced theory that considers the delocalization of electrons over the entire molecule, render a more accurate description of tie.
- Steric Effects: The spatial arrangement of atoms and groups in a molecule, which can influence reactivity and constancy.
These advanced topics provide a more nuanced understanding of molecular behavior and are essential for consider complex systems.
for instance, view the molecule CH2O (formaldehyde):
- The central carbon atom is sp2 hybridized, forming a rhombohedral planar geometry.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, add to the molecule's constancy.
Understanding these boost concepts can facilitate in augur the doings of more complex molecules and designing new materials with specific properties.
for representative, study the molecule C6H6 (benzene):
- The carbon atoms are sp2 interbreed, make a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, contributing to the molecule's aromaticity and constancy.
These advanced topics cater a more nuanced understanding of molecular behaviour and are all-important for studying complex systems.
for illustration, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridise, form a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and constancy.
Understanding these supercharge concepts can help in call the doings of more complex molecules and designing new materials with specific properties.
for representative, view the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridize, organise a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and stability.
These advanced topics provide a more nuanced understanding of molecular doings and are essential for studying complex systems.
for instance, deal the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, constitute a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and stability.
Understanding these advanced concepts can help in predict the behavior of more complex molecules and designing new materials with specific properties.
for example, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, organize a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, contributing to the molecule's aromaticity and stability.
These advance topics render a more nuanced read of molecular demeanor and are crucial for analyze complex systems.
for illustration, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 interbreed, organise a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and stability.
Understanding these progress concepts can help in foreshadow the behavior of more complex molecules and project new materials with specific properties.
for case, take the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, forming a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, lend to the molecule's aromaticity and constancy.
These supercharge topics furnish a more nuanced understanding of molecular behavior and are essential for examine complex systems.
for instance, regard the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, constitute a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, add to the molecule's aromaticity and constancy.
Understanding these advanced concepts can help in predicting the behavior of more complex molecules and plan new materials with specific properties.
for instance, view the molecule C6H6 (benzene):
- The carbon atoms are sp2 crossbreed, organize a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and constancy.
These advance topics furnish a more nuanced understanding of molecular behavior and are essential for studying complex systems.
for illustration, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 interbreed, forming a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, contributing to the molecule's aromaticity and constancy.
Understanding these advanced concepts can aid in augur the conduct of more complex molecules and design new materials with specific properties.
for instance, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, forming a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, bring to the molecule's aromaticity and stability.
These advanced topics render a more nuanced see of molecular behavior and are essential for studying complex systems.
for instance, take the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, constitute a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, bestow to the molecule's aromaticity and constancy.
Understanding these advanced concepts can aid in predicting the behavior of more complex molecules and contrive new materials with specific properties.
for instance, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 crossbreed, organise a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, add to the molecule's aromaticity and stability.
These supercharge topics provide a more nuanced translate of molecular behaviour and are crucial for consider complex systems.
for example, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, make a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and constancy.
Understanding these supercharge concepts can facilitate in predicting the behavior of more complex molecules and contrive new materials with specific properties.
for representative, take the molecule C6H6 (benzene):
- The carbon atoms are sp2 crossbreed, organise a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, give to the molecule's aromaticity and stability.
These advanced topics ply a more nuanced realize of molecular demeanour and are crucial for study complex systems.
for instance, see the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridise, make a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, bring to the molecule's aromaticity and constancy.
Understanding these progress concepts can help in predicting the behaviour of more complex molecules and contrive new materials with specific properties.
for instance, take the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridize, forming a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, contribute to the molecule's aromaticity and stability.
These supercharge topics provide a more nuanced understanding of molecular behavior and are crucial for studying complex systems.
for instance, regard the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, forming a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, contributing to the molecule's aromaticity and stability.
Understanding these advanced concepts can help in predicting the behavior of more complex molecules and designing new materials with specific properties.
for instance, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, forming a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, lead to the molecule's aromaticity and constancy.
These boost topics cater a more nuanced interpret of molecular behavior and are indispensable for studying complex systems.
for case, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridize, organize a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, bestow to the molecule's aromaticity and stability.
Understanding these advance concepts can assist in predicting the behavior of more complex molecules and design new materials with specific properties.
for example, reckon the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, form a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, conduce to the molecule's aromaticity and constancy.
These advanced topics furnish a more nuanced realize of molecular behavior and are all-important for canvas complex systems.
for instance, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridise, forming a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π scheme, contributing to the molecule's aromaticity and stability.
Understanding these advance concepts can facilitate in prefigure the behavior of more complex molecules and designing new materials with specific properties.
for example, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 crossbreed, forming a planar ring structure.
- The molecular orbital theory explains the delocalization of electrons in the π system, bestow to the molecule's aromaticity and stability.
These boost topics cater a more nuanced realise of molecular doings and are all-important for study complex systems.
for representative, consider the molecule C6H6 (benzene):
- The carbon atoms are sp2 hybridized, make a planar ring construction.
- The molecular orbital theory explains the delocalization of electrons in the π system, contributing to the molecule's aromaticity and stability.
Understanding these advanced concepts can help in predicting the deportment of more complex molecules and designing new materials with specific properties.
for instance, consider the molecule C6H6 (benzene):
- The
Related Terms:
- molecular vs electron pair geometry
- electron and molecular geometry quizlet
- electron geometry vs molecular shape
- electron geometry vs molecular examples
- 5 electron domain geometry
- molecular geometry vs electron domain