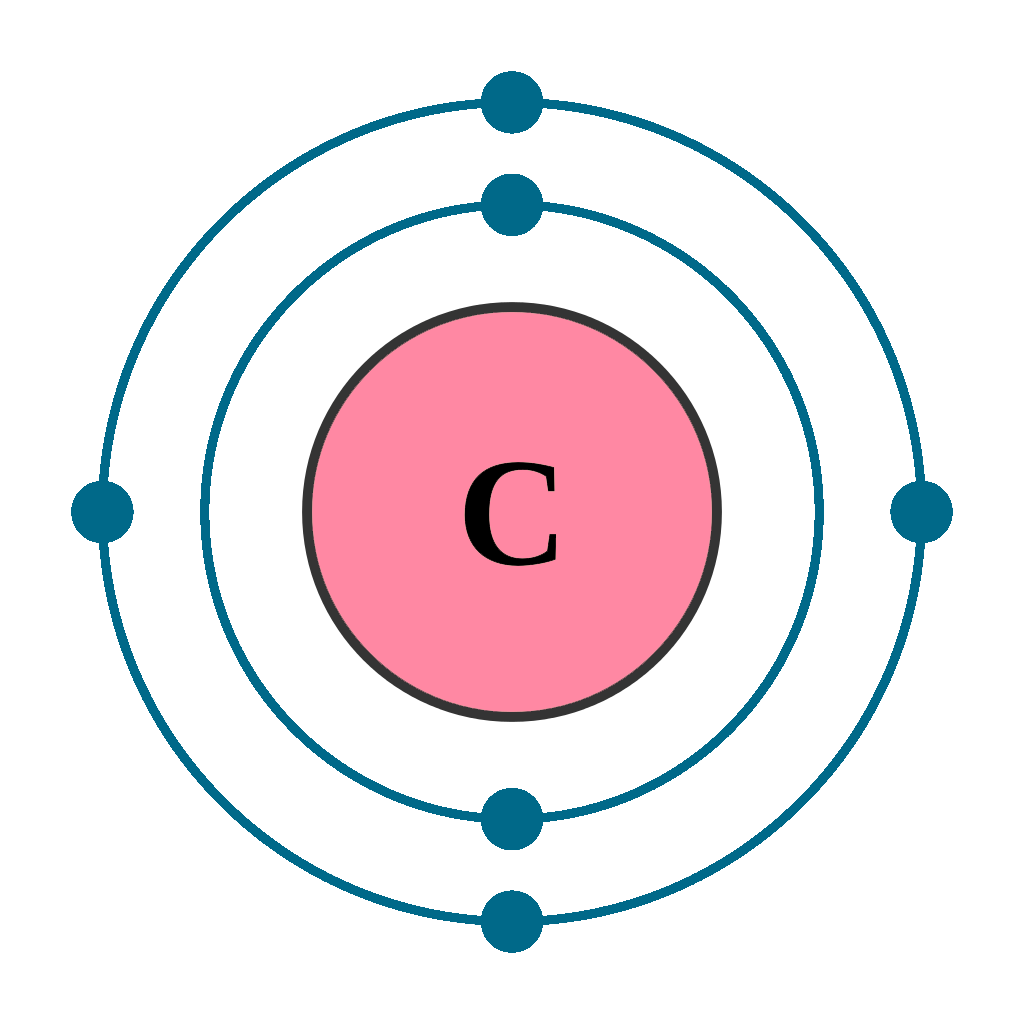
Understanding the electron distribution of carbon is key to comprehend the behavior of this essential element in assorted chemical reactions and materials. Carbon, with its atomic number 6, has a unequalled electron configuration that allows it to form a broad array of compounds, get it a cornerstone of organic chemistry and materials science.
Electron Configuration of Carbon
Carbon has six electrons, which are distributed across its nuclear orbitals. The electron constellation of carbon is 1s 2 2s2 2p2. This configuration can be break down as follows:
- 1s orbital: Contains 2 electrons.
- 2s orbital: Contains 2 electrons.
- 2p orbital: Contains 2 electrons.
The 2p orbital is particularly important because it has three sub orbitals (2p x, 2p y, and 2p z ), each of which can hold up to two electrons. In the ground state, carbon has two electrons in the 2p orbital, but these electrons can be excited to higher energy levels, allowing carbon to form various types of bonds.
Bonding and Hybridization
The electron dispersion of carbon enables it to form potent covalent bonds with other atoms. Carbon's power to form single, double, and triple bonds is due to its crossbreeding, a concept that explains the immix of nuclear orbitals to form new hybrid orbitals. The most mutual types of hybridizing in carbon are:
- sp 3 crossbreeding: Occurs when one s orbital and three p orbitals mix to form four sp 3 hybrid orbitals. This type of hybridization is common in alkanes, where carbon forms single bonds with four other atoms.
- sp 2 hybridization: Involves one s orbital and two p orbitals mixing to form three sp 2 hybrid orbitals. This is distinctive in alkenes, where carbon forms double bonds.
- sp hybridization: Results from one s orbital and one p orbital meld to form two sp hybrid orbitals. This type of cross is found in alkynes, where carbon forms triple bonds.
These hybridizations let carbon to form a variety of molecular geometries, include tetrahedral (sp 3 ), trigonal planar (sp2 ), and linear (sp).
Carbon Allotropes
The electron dispersion of carbon also plays a crucial role in the shaping of different allotropes of carbon, which are distinct forms of the element with different physical properties. The most easily known allotropes are:
- Diamond: In diamond, each carbon atom is sp 3 hybridize and forms four potent covalent bonds with neighboring carbon atoms, lead in a extremely rigid and potent construction.
- Graphite: In graphite, carbon atoms are sp 2 hybridized and stage in hexagonal layers. Each carbon atom forms three covalent bonds with neighboring atoms, and the layers are held together by weak van der Waals forces, grant them to slide over each other.
- Fullerenes: Fullerenes, such as C 60 (buckminsterfullerene), have a orbicular or ellipsoidal construction composed of carbon atoms arranged in a cage like fashion. The carbon atoms are sp 2 hybridized, and the structure is brace by delocalize π electrons.
- Carbon Nanotubes: These are cylindric structures made of sp 2 hybridized carbon atoms. They can be single fence or multi wall and have unique electric and mechanical properties.
- Graphene: Graphene is a single layer of graphite, consisting of sp 2 hybridized carbon atoms arranged in a hexagonal lattice. It has exceptional strength, conductivity, and flexibility.
Each of these allotropes has unparalleled properties due to the different arrangements and hybridizations of carbon atoms, which are directly influenced by the electron distribution of carbon.
Carbon in Organic Chemistry
Carbon's power to form a vast array of compounds is due to its electron distribution, which allows it to form stable covalent bonds with other carbon atoms and with atoms of other elements. This versatility is the basis of organic chemistry, the study of carbon containing compounds. Some key points about carbon in organic chemistry include:
- Catenation: Carbon's ability to form strong bonds with other carbon atoms allows for the shaping of long chains and rings, a property known as catenation.
- Functional Groups: Carbon can bond with various functional groups, such as hydroxyl (OH), carbonyl (C O), carboxyl (COOH), and amino (NH 2 ), to form different types of organic compounds.
- Isomerism: The electron dispersion of carbon allows for the existence of isomers, compounds with the same molecular formula but different structures and properties.
These properties create carbon the backbone of organic chemistry, enabling the formation of complex molecules all-important for life and several industrial applications.
Carbon in Materials Science
The electron distribution of carbon also plays a significant role in materials skill, where carbon establish materials are used in a wide range of applications. Some far-famed examples include:
- Diamond: Used in swerve tools, abrasives, and high execution electronics due to its callosity and caloric conduction.
- Graphite: Employed in pencils, lubricants, and electrodes in batteries and fuel cells.
- Fullerenes: Used in materials science for their unique electronic and mechanical properties, as well as in medical applications for drug delivery.
- Carbon Nanotubes: Utilized in electronics, materials reinforcement, and energy storage due to their exceptional strength and conductivity.
- Graphene: Applied in electronics, sensors, and composite materials for its eminent conductivity, strength, and tractability.
These applications spotlight the importance of interpret the electron distribution of carbon in evolve advanced materials with sew properties.
Electron Distribution and Chemical Reactivity
The electron distribution of carbon importantly influences its chemic reactivity. Carbon's power to form multiple bonds and its tendency to achieve a stable octet form make it highly responsive. Some key aspects of carbon's reactivity include:
- Radical Formation: Carbon can form radicals, which are highly reactive species with unpaired electrons. These radicals play a crucial role in many chemical reactions, including polymerization and combustion.
- Electrophilic and Nucleophilic Substitution: Carbon atoms in organic compounds can undergo electrophilic and nucleophilic substitution reactions, where an electrophile or nucleophile replaces an exist group on the carbon atom.
- Addition Reactions: Carbon carbon double and triple bonds can undergo add-on reactions, where reactants add across the multiple bond to form new compounds.
Understanding these reactivity patterns is indispensable for predicting and operate chemic reactions imply carbon compounds.
Note: The reactivity of carbon is also tempt by the presence of other atoms and functional groups in the molecule, which can affect the electron distribution and stability of the compound.
Carbon in Biological Systems
Carbon is the fundamental building block of life, and its electron distribution enables the constitution of complex biomolecules essential for biologic processes. Some key biologic molecules containing carbon include:
- Proteins: Composed of amino acids linked by peptide bonds, proteins play crucial roles in structural indorse, enzymatic action, and cellular point.
- Nucleic Acids: DNA and RNA are polymers of nucleotides, which curb carbon based sugars, phosphate groups, and nitrogen-bearing bases. These molecules store and transmit transmissible information.
- Carbohydrates: Sugars and polysaccharides, such as glucose and cellulose, are carbon based molecules that serve as energy sources and structural components in living organisms.
- Lipids: Fats, oils, and waxes are carbon free-base molecules that store energy, ply insularity, and form cellular membranes.
The electron distribution of carbon allows these biomolecules to form stable structures and interact with each other in complex biologic processes.
Carbon in Environmental Science
The electron distribution of carbon also plays a critical role in environmental skill, specially in the context of the carbon cycle and climate change. Carbon is invariably convert between the atmosphere, biosphere, hydrosphere, and lithosphere through respective processes, include:
- Photosynthesis: Plants and other photosynthetic organisms convert carbon dioxide into organic compounds using sunlight energy.
- Respiration: Organisms break down organic compounds to release energy, producing carbon dioxide as a byproduct.
- Combustion: The burning of fossil fuels and biomass releases carbon dioxide into the atmosphere.
- Weathering and Erosion: Carbon contain minerals and rocks are separate down and enthral through geologic processes.
Understanding the electron distribution of carbon and its role in these processes is all-important for addressing environmental challenges, such as climate change and carbon sequestration.
Carbon s unequalled electron dispersion makes it a versatile and essential element in several scientific disciplines. From its role in organic chemistry and materials skill to its importance in biologic systems and environmental processes, carbon s properties are key to our understand of the natural world. By canvas the electron distribution of carbon, scientists can evolve new materials, meliorate chemical reactions, and address environmental challenges, ultimately supercharge our knowledge and technology.
Related Terms:
- carbon electron configuration diagram
- complete electron conformation of carbon
- electron ring diagram for carbon
- electron distribution diagram for carbon
- carbon electron shell diagram
- total electron configuration of carbon