Understanding the electronic configuration of elements is key to comprehend their chemical properties and behaviors. Among the elements, manganese (Mn) stands out due to its alone electronic contour and various applications. This blog post delves into the electronic constellation of Mn, its meaning, and its role in various chemic and industrial processes.
What is Electronic Configuration?
Electronic shape refers to the arrangement of electrons in the orbitals of an atom. This arrangement determines the chemic properties of an element, include its reactivity, bonding behavior, and electronic structure. The electronic configuration is typically represented using the notation that specifies the number of electrons in each orbital.
Electronic Configuration of Manganese (Mn)
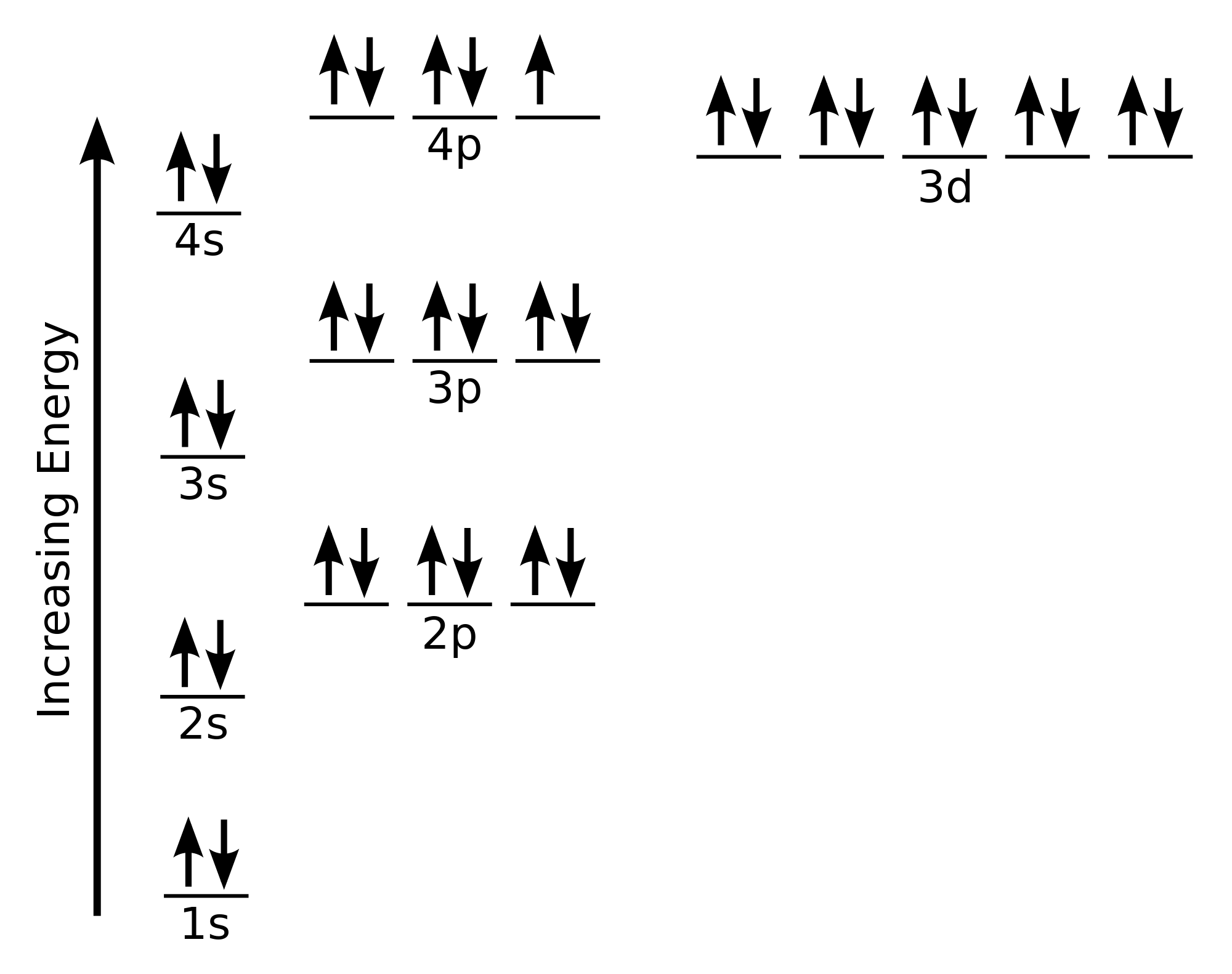
Manganese (Mn) is a passage metallic with the atomic number 25. Its electronic configuration is important for understanding its chemical demeanor. The electronic configuration of Mn is:
1s 2 2s 2 2p 6 3s 2 3p 6 3d 5 4s 2
This contour indicates that Mn has:
- 2 electrons in the 1s orbital
- 2 electrons in the 2s orbital
- 6 electrons in the 2p orbital
- 2 electrons in the 3s orbital
- 6 electrons in the 3p orbital
- 5 electrons in the 3d orbital
- 2 electrons in the 4s orbital
The 3d and 4s orbitals are particularly significant in the chemistry of changeover metals like Mn. The 3d orbital can hold up to 10 electrons, and the 4s orbital can hold up to 2 electrons. The presence of electrons in these orbitals contributes to the unique properties of Mn, include its variable oxidation states and catalytic activity.
Significance of the Electronic Configuration of Mn
The electronic configuration of Mn plays a polar role in its chemic and physical properties. Some key points to view are:
- Variable Oxidation States: Mn can exhibit multiple oxidation states, vagabond from 3 to 7. This versatility is due to the power of its 3d and 4s electrons to participate in chemical attach. The most common oxidation states are 2, 3, 4, 6, and 7.
- Catalytic Activity: The electronic configuration of Mn allows it to act as a catalyst in respective chemical reactions. Its power to alter oxidation states makes it effectual in facilitate redox reactions.
- Magnetic Properties: The front of unpaired electrons in the 3d orbital contributes to the magnetized properties of Mn. This makes Mn useful in applications requiring magnetic materials.
- Biological Importance: Mn is an essential trace element in biological systems. It plays a role in various enzymatic reactions and is imply in processes such as photosynthesis and respiration.
Applications of Manganese
The unique electronic configuration of Mn makes it valuable in numerous applications across various industries. Some of the key applications include:
- Steel Production: Mn is widely used in the product of steel to improve its strength, callosity, and resistance to wear and tear. It is added to steel in the form of ferroalloys.
- Batteries: Mn is used in the production of alkaline and lithium ion batteries. Its power to undergo redox reactions makes it an efficacious component in battery electrodes.
- Catalysis: Mn based catalysts are used in various chemic processes, including the production of chemicals, pharmaceuticals, and fuels. Its catalytic properties are leverage to heighten response rates and selectivity.
- Agriculture: Mn is an essential nutrient for plants and is used in fertilizers to promote growth and development. It plays a role in photosynthesis and enzyme energizing.
- Electronics: Mn is used in the product of electronic components, such as capacitors and resistors. Its magnetised properties make it utile in the manufacturing of magnetized storage devices.
Chemical Properties of Manganese
The electronic shape of Mn influences its chemical properties, making it a versatile element in chemical reactions. Some key chemic properties include:
- Reactivity: Mn is a reactive metallic that can form compounds with various elements. It readily reacts with oxygen to form manganese oxides, which are used in ceramics and glass product.
- Oxidation States: As mentioned earlier, Mn can exhibit multiple oxidation states. This property is important in its role as a catalyst and in its power to form a encompassing range of compounds.
- Complex Formation: Mn can form complex compounds with ligands, such as h2o, ammonia, and organic molecules. These complexes are crucial in catalysis and biologic systems.
- Redox Reactions: Mn s ability to undergo redox reactions makes it useful in diverse chemic processes. It can act as both an oxidizing and cut agent, count on the reaction conditions.
Environmental and Health Considerations
While Mn is essential for various industrial and biological processes, it also poses environmental and health considerations. Understanding its electronic configuration helps in managing its use and disposal:
- Environmental Impact: Mn mining and process can lead to environmental pollution, include soil and h2o contamination. Proper management of waste materials is crucial to denigrate environmental wallop.
- Health Effects: Exposure to eminent levels of Mn can have adverse health effects, include neurologic disorders. Workers in industries affect Mn should take conquer safety measures to protect against exposure.
- Regulation and Safety: Regulations and safety guidelines are in place to handle the use of Mn in various applications. These guidelines assist secure that Mn is used safely and responsibly, minimizing risks to both the environment and human health.
Note: Always postdate safety protocols when handling manganese and its compounds to avoid likely health risks.
Manganese s electronic shape is a cornerstone of its chemic and physical properties, do it a valuable element in various applications. From steel product to catalysis and biologic systems, the unequalled arrangement of electrons in Mn s orbitals contributes to its versatility and importance. Understanding the electronic configuration of Mn provides insights into its doings and helps in optimise its use in different industries. By leveraging its properties, we can continue to develop innovative solutions and technologies that benefit society.
Related Terms:
- entire electron configuration for mn
- electronic configuration of mn2
- electronic configuration of ni
- electronic configuration of mn 3
- complete electron configuration for manganese
- electronic conformation of manganese 25