Understanding the molecular geometry of compounds is key in chemistry, as it helps predict their properties and behaviors. One such compound that exhibits intriguing molecular geometry is sulfur tetrafluoride (SF 4 ). This compound is a fascinating subject for study due to its unique structure and the insights it provides into the principles of molecular geometry.
Introduction to Molecular Geometry
Molecular geometry refers to the three dimensional arrangement of atoms in a molecule. This arrangement is crucial because it determines the molecule s physical and chemic properties. The geometry of a molecule is influenced by various factors, include the bit of electron pairs around the central atom, the type of bonds (single, double, triple), and the presence of lone pairs.
The Structure of Sulfur Tetrafluoride (SF 4 )
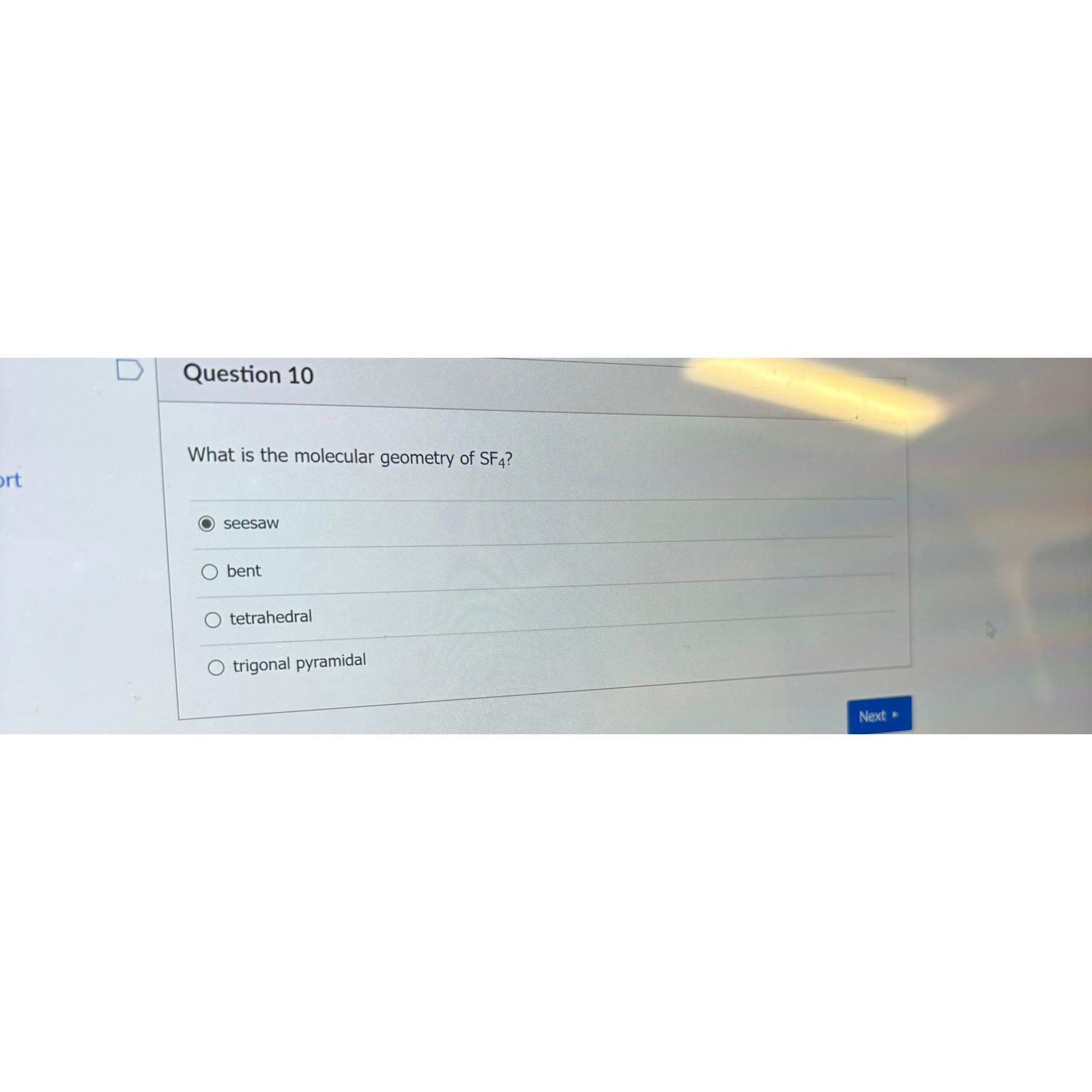
Sulfur tetrafluoride is a chemical compound with the formula SF 4. It consists of one sulfur atom bonded to four fluorine atoms. The molecular geometry of SF 4 is a seesaw shape, which is a bit strange equate to more mutual geometries like tetrahedral or trigonal bipyramidal.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
The VSEPR theory is a powerful instrument for predicting the molecular geometry of compounds. According to this theory, electron pairs around a central atom repel each other and arrange themselves in a way that minimizes standoff. For SF 4, the sulfur atom has five electron pairs: four adhere pairs and one lone pair.
To understand the molecular geometry of SF 4, let's break down the steps imply in apply VSEPR theory:
- Identify the fundamental atom: In SF 4, the primal atom is sulfur (S).
- Count the total number of valence electrons: Sulfur has 6 valency electrons, and each fluorine atom contributes 7 valency electrons. Therefore, the full turn of valency electrons is 6 (from S) 4 7 (from F) 34.
- Determine the number of attach and lone pairs: There are four S F bonds, which account for 8 electrons. The remaining 26 electrons are dispense as lone pairs on the fluorine atoms. The sulfur atom has one lone pair.
- Arrange the electron pairs: The five electron pairs around the sulfur atom will arrange themselves in a rhombohedral bipyramidal geometry to minimize repulsion.
- Consider the lone pair: The presence of a lone pair on the sulfur atom distorts the rhombohedral bipyramidal geometry, resulting in a seesaw shape.
Note: The lone pair on the sulfur atom occupies more space than the bonding pairs, starring to a greater revulsion and a deformation from the ideal rhombohedral bipyramidal geometry.
Seesaw Molecular Geometry
The seesaw molecular geometry of SF 4 can be visualize as follows:
Imagine a trigonal bipyramidal arrangement where one of the axial positions is occupied by a lone pair. The two axile positions are at 180 degrees to each other, and the three equatorial positions are at 120 degrees to each other. The lone pair pushes the axile fluorine atoms closer together, ensue in a seesaw shape.
Here is a table summarizing the key points of the seesaw molecular geometry:
| Central Atom | Number of Bonding Pairs | Number of Lone Pairs | Geometry |
|---|---|---|---|
| Sulfur (S) | 4 | 1 | Seesaw |
Properties of Sulfur Tetrafluoride
The unique molecular geometry of SF 4 influences its physical and chemic properties. Some key properties include:
- Molecular Shape: Seesaw, with a lone pair on the sulfur atom.
- Bond Angles: The bond angles are falsify due to the lone pair, with the axial F S F angle being less than 180 degrees and the equatorial F S F angles being less than 120 degrees.
- Polarity: SF 4 is a polar molecule due to the front of the lone pair and the asymmetrical dispersion of electron concentration.
- Reactivity: Sulfur tetrafluoride is highly responsive and can act as a fluorinating agent in chemical reactions.
Applications of Sulfur Tetrafluoride
Despite its reactivity, SF 4 has several important applications in chemistry and industry. Some of these applications include:
- Fluorination Reactions: SF 4 is used as a fluorinating agent in the synthesis of various organic and inorganic compounds.
- Catalysts: It is used in the provision of catalysts for certain chemic reactions.
- Research: SF 4 is studied in academic research to read the principles of molecular geometry and chemic bonding.
Understanding the molecular geometry of SF 4 provides valuable insights into the doings of molecules with lone pairs and the distortions they get in molecular shapes. This noesis is essential for predicting the properties and reactivity of similar compounds.
In compact, the molecular geometry of SF 4 is a authoritative illustration of how the front of lone pairs can distort the ideal geometry predicted by VSEPR theory. The seesaw shape of SF 4 results from the horror between the lone pair and the bonding pairs, leading to unequalled properties and applications. By studying the molecular geometry of SF 4, chemists gain a deeper understanding of the factors that influence molecular shape and behaviour.
Related Terms:
- electron geometry of sf4
- molecular geometry of co2
- sf4 lewis construction
- molecular geometry of h2o
- bond angles of sf4
- molecular geometry of clf3