Understanding the Scn Lewis Structure is fundamental for anyone dig into the existence of chemistry, particularly in the study of molecular bind and electron distribution. The Lewis structure, named after Gilbert N. Lewis, provides a unproblematic yet knock-down way to fancy the attach between atoms in a molecule. This structure helps chemists predict the geometry, reactivity, and other properties of molecules. In this post, we will explore the basics of the Scn Lewis Structure, its significance, and how to draw it step by step.
What is a Lewis Structure?
A Lewis construction, also known as a Lewis dot diagram or electron dot construction, is a diagrammatical representation of the valence electrons in a molecule. It shows how atoms are bonded together and how electrons are spread around them. The structure consists of dots correspond valency electrons and lines typify covalent bonds.
Importance of Lewis Structures
Lewis structures are crucial for various reasons:
- Predicting Molecular Geometry: They help in portend the three dimensional shape of a molecule, which is indispensable for understanding its properties.
- Understanding Reactivity: By showing the distribution of electrons, Lewis structures can indicate which parts of a molecule are more potential to react with other molecules.
- Identifying Bond Types: They differentiate between single, double, and triple bonds, which is crucial for translate the strength and nature of the bonds.
- Explaining Chemical Properties: Lewis structures provide insights into the chemical properties of molecules, such as polarity and acidity.
Drawing a Lewis Structure
Drawing a Scn Lewis Structure involves several steps. Let s go through the process using the exemplar of the sulfur dioxide (SO2) molecule.
Step 1: Determine the Total Number of Valence Electrons
The first step is to count the full routine of valency electrons in the molecule. Valence electrons are the electrons in the outermost shell of an atom. For SO2:
- Sulfur (S) has 6 valency electrons.
- Oxygen (O) has 6 valence electrons each.
So, the entire number of valency electrons for SO2 is:
6 (from S) 6 (from O) 6 (from O) 18 valence electrons.
Step 2: Choose the Central Atom
The fundamental atom is usually the least electronegative atom in the molecule. In SO2, sulfur (S) is the key atom because it is less negative than oxygen (O).
Step 3: Connect the Atoms with Single Bonds
Draw single bonds between the cardinal atom and the surrounding atoms. For SO2, this means drawing single bonds between sulfur and each oxygen atom.
Step 4: Distribute the Remaining Electrons
After connecting the atoms with single bonds, distribute the remaining valency electrons around the atoms to satisfy the octet rule. The octet rule states that atoms tend to have eight electrons in their valence shell to achieve constancy.
For SO2, after delineate the single bonds, we have used 4 electrons (2 for each bond). This leaves us with 14 electrons to distribute. Place these electrons around the oxygen atoms first, as they are more negative and need to complete their octet.
Step 5: Form Multiple Bonds if Necessary
If any atom does not have an octet, form multiple bonds by go lone pairs of electrons from surrounding atoms to form double or triple bonds. In SO2, each oxygen atom needs 2 more electrons to complete its octet. Move one lone pair from each oxygen atom to form double bonds with the sulfur atom.
Step 6: Check for Formal Charges
Calculate the formal charge on each atom to check the structure is correct. The formal charge is calculated as:
Formal Charge (Number of valency electrons in costless atom) (Number of lone pair electrons) (Number of adhere electrons 2)
For SO2, the formal charges should be zero for a stable structure. If there are formal charges, adjust the construction by moving electrons to belittle them.
Example of a Scn Lewis Structure
Let s draw the Scn Lewis Structure for sulfur dioxide (SO2) step by step:
1. Total valence electrons: 18
2. Central atom: Sulfur (S)
3. Connect with single bonds:

4. Distribute remaining electrons:

5. Form multiple bonds:

6. Check formal charges:
Sulfur: 6 2 4 0
Oxygen: 6 6 1 1
Oxygen: 6 6 1 1
To minimise formal charges, we can move electrons to form a double bond with one oxygen and a single bond with the other, resulting in a stable construction with zero formal charges.
Note: The Scn Lewis Structure for SO2 can also be drawn with sonority structures to wagerer typify the electron distribution.
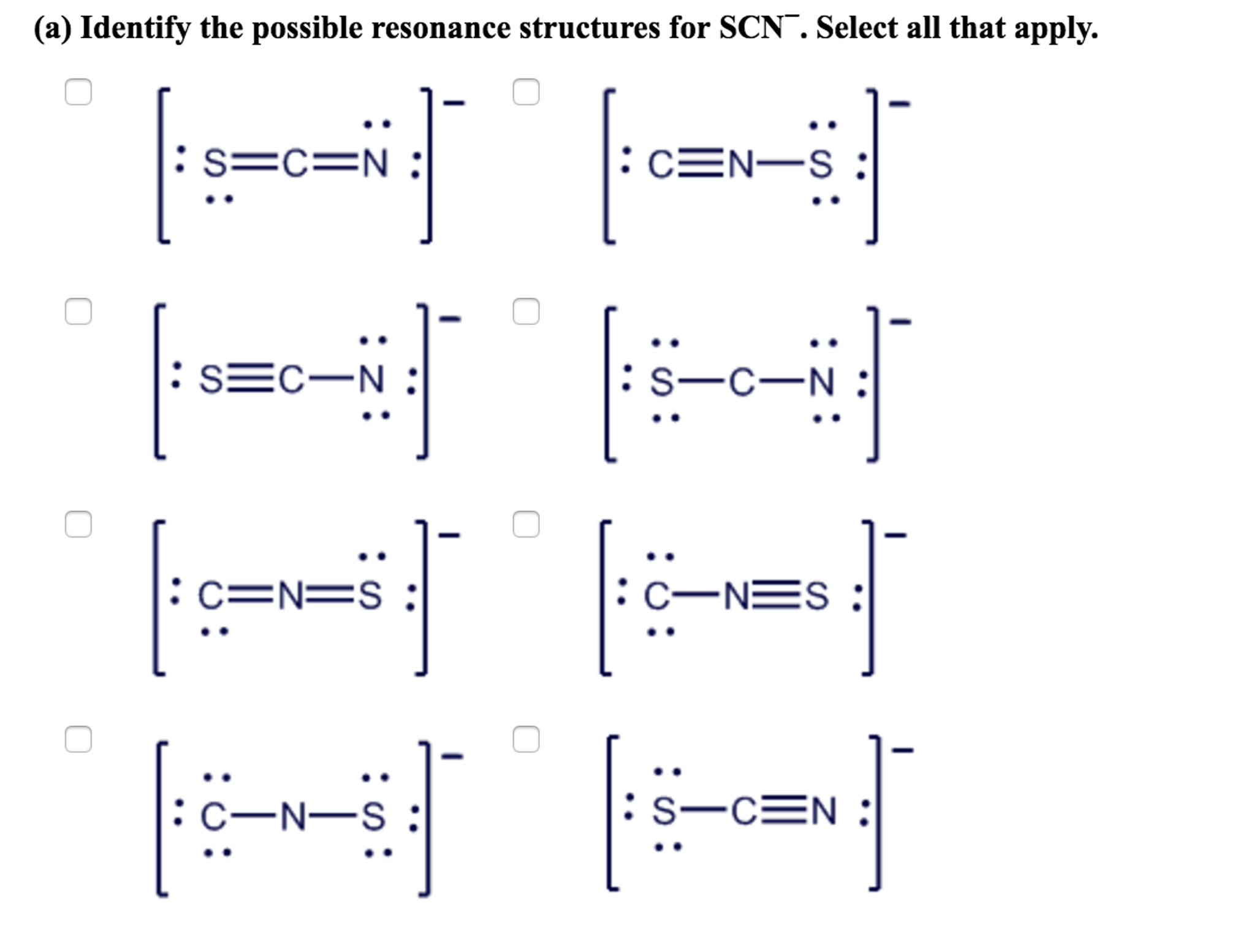
Resonance Structures
Resonance structures are different Lewis structures of the same molecule that differ only in the position of electrons. They are used to symbolise molecules where a single Lewis structure cannot accurately describe the electron distribution. For SO2, the resonance structures assist show the delocalization of electrons between the sulfur and oxygen atoms.
Resonance structures for SO2:

Exceptions to the Octet Rule
While the octet rule is a useful guideline, there are exceptions where atoms do not have eight electrons in their valency shell. These exceptions include:
- Molecules with an Odd Number of Electrons: Examples include nitric oxide (NO) and chlorine dioxide (ClO2).
- Molecules with Less than an Octet: Examples include boron trifluoride (BF3) and beryllium chloride (BeCl2).
- Molecules with More than an Octet: Examples include sulfur hexafluoride (SF6) and phosphorus pentafluoride (PF5). These molecules have expanded valence shells due to the front of d orbitals.
Formal Charge Calculation
Formal charge is a concept used to determine the most stable Lewis construction. It is compute using the formula:
Formal Charge (Number of valency electrons in free atom) (Number of lone pair electrons) (Number of adhere electrons 2)
Let s calculate the formal charges for the atoms in the Scn Lewis Structure of SO2:
| Atom | Valence Electrons | Lone Pair Electrons | Bonding Electrons | Formal Charge |
|---|---|---|---|---|
| Sulfur (S) | 6 | 2 | 4 | 0 |
| Oxygen (O) | 6 | 6 | 2 | 1 |
| Oxygen (O) | 6 | 6 | 2 | 1 |
To minimize formal charges, adjust the construction to ascertain that the formal charges on all atoms are as close to zero as possible.
Note: The Scn Lewis Structure with the lowest formal charges is broadly the most stable.
Practice Examples
To master delineate Scn Lewis Structures, practice with various molecules. Here are a few examples to get you commence:
- Carbon dioxide (CO2)
- Water (H2O)
- Ammonia (NH3)
- Methane (CH4)
- Nitrogen dioxide (NO2)
For each molecule, follow the steps adumbrate earlier to draw the Lewis structure and check for formal charges.
By practicing with different molecules, you will become proficient in line Scn Lewis Structures and read the bond and electron distribution in several compounds.
Lewis structures are a primal creature in chemistry, provide insights into molecular geometry, reactivity, and properties. By mastering the art of drawing Scn Lewis Structures, you will gain a deeper understanding of chemic bond and the doings of molecules. This knowledge is essential for further studies in chemistry and relate fields.
Related Terms:
- scn resonance
- clcn lewis construction
- scn lewis structure diagram
- scn formal charge
- scn interbreeding
- n3 lewis structure