Understanding the concept of sites of unsaturation is essential in the battlefield of organic chemistry. These sites refer to the locations within a molecule where double or triple bonds are present, make them highly responsive. This reactivity is due to the front of π (pi) bonds, which are more susceptible to chemical attacks compared to σ (sigma) bonds. By exploring the properties and behaviors of these sites, chemists can predict and control chemic reactions, star to the synthesis of new compounds with trust properties.
What are Sites of Unsaturation?
Sites of unsaturation are regions in a molecule where double or triple bonds exist. These bonds are qualify by the front of π electrons, which are delocalized and can participate in various chemic reactions. The most mutual types of unsaturation include:
- Alkenes: Contain a carbon carbon double bond (C C).
- Alkynes: Contain a carbon carbon triple bond (C C).
- Aromatic compounds: Contain delocalize π electrons in a ring construction, such as benzene.
These sites are particularly important in organic synthesis, as they often function as starting points for reactions that can introduce new functional groups or qualify exist ones.
Identifying Sites of Unsaturation
Identifying sites of unsaturation in a molecule is a rudimentary skill in organic chemistry. There are several methods to shape these sites, including:
- Spectroscopic Techniques: Infrared (IR) spectroscopy and Nuclear Magnetic Resonance (NMR) spectroscopy are commonly used to identify the presence of double or triple bonds.
- Chemical Tests: Reagents like bromine (Br2) and potassium permanganate (KMnO4) can react with unsaturated sites, providing optical indicators of their presence.
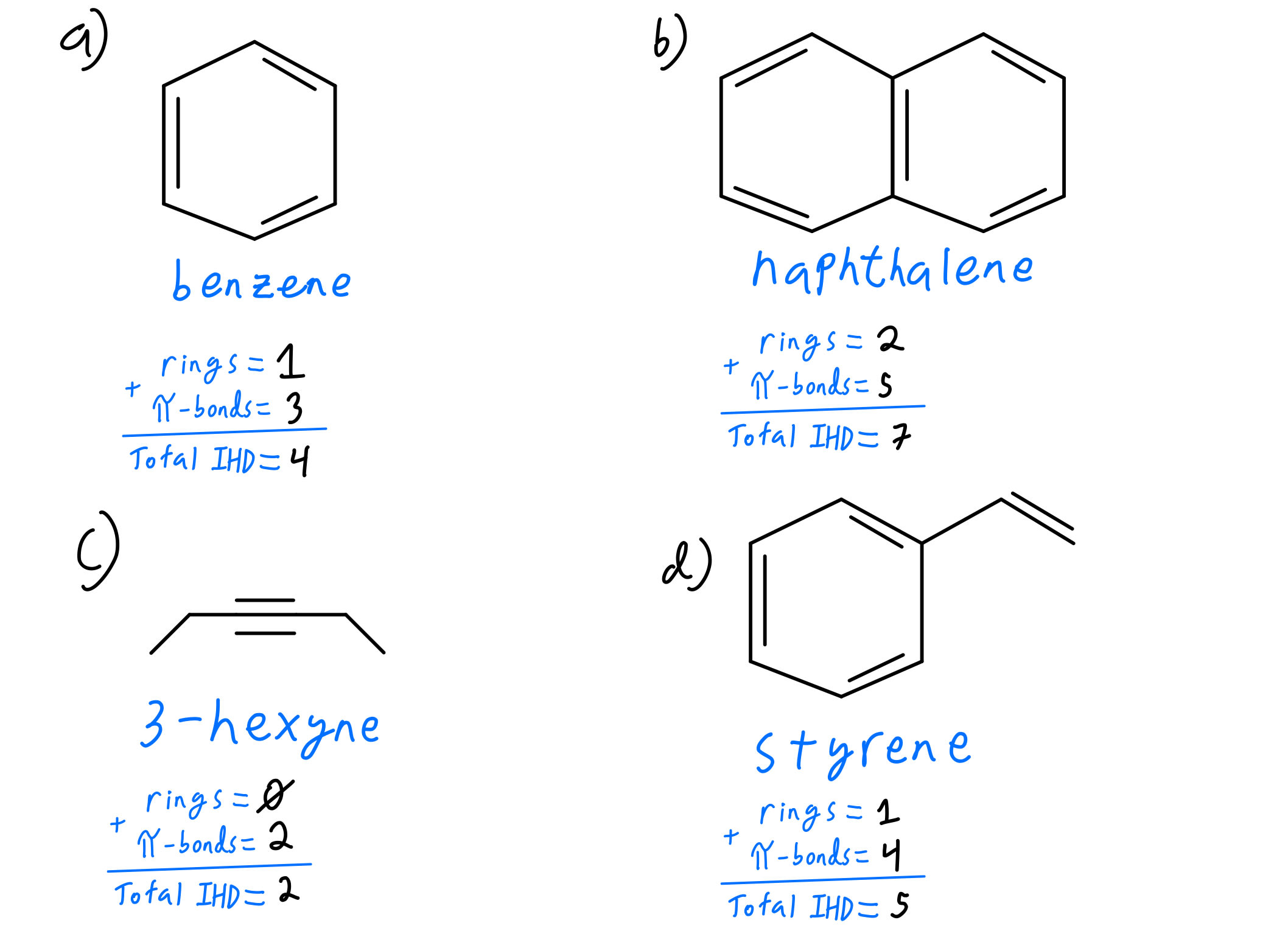
- Degree of Unsaturation: This can be calculated using the formula: (2C 2 N X H) 2, where C is the number of carbon atoms, N is the number of nitrogen atoms, X is the number of halogen atoms, and H is the act of hydrogen atoms. This formula helps ascertain the turn of rings and π bonds in a molecule.
Reactivity of Sites of Unsaturation
The reactivity of sites of unsaturation is principally due to the front of π electrons, which are more broadly held compare to σ electrons. This makes them more susceptible to electrophilic and nucleophilic attacks. Some key reactions involve these sites include:
- Electrophilic Addition: In this reaction, an electrophile (electron loving species) attacks the π bond, leading to the formation of a new bond. for instance, the gain of hydrogen bromide (HBr) to an alkene results in the formation of an alkyl halide.
- Nucleophilic Addition: In this reaction, a nucleophile (electron rich species) attacks the π bond, leading to the formation of a new bond. for instance, the addition of cyanide (CN) to an alkene results in the formation of a nitrile.
- Oxidation: Unsaturated sites can be oxidize using reagents like potassium permanganate (KMnO4) or osmium tetroxide (OsO4). This reaction can convert alkenes to diols or alkynes to carboxyl acids.
Applications of Sites of Unsaturation
The reactivity of sites of unsaturation makes them worthful in various applications, including:
- Pharmaceuticals: Many drugs bear unsaturated sites, which are crucial for their biologic activity. for illustration, the double bond in aspirin is indispensable for its anti inflammatory properties.
- Polymers: Unsaturated sites are used in the synthesis of polymers, such as polyethylene and polypropylene. The double bonds in these monomers let for polymerization reactions, leading to the formation of long chain polymers.
- Natural Products: Many natural products, such as terpenes and steroids, bear unsaturated sites. These sites are frequently involved in the biosynthesis of these compounds and their biological activities.
Examples of Sites of Unsaturation
To bettor interpret sites of unsaturation, let s look at some examples:
| Compound | Structure | Sites of Unsaturation |
|---|---|---|
| Ethene | CH2 CH2 | One double bond |
| Ethyne | HC CH | One triple bond |
| Benzene |

|
Three delocalized π bonds |
Note: The structure of benzene is oftentimes typify as a hexagon with jump single and double bonds, but in reality, it has delocalized π electrons.
Challenges and Considerations
While sites of unsaturation offer legion opportunities for chemical reactions, they also demonstrate challenges. Some key considerations include:
- Selectivity: Ensuring that reactions occur at the hope site of unsaturation can be dispute, especially in molecules with multiple unsaturated sites.
- Stability: Unsaturated sites can be unstable and prone to abasement, specially in the front of air or light.
- Stereochemistry: The geometry of unsaturated sites can involve the stereochemistry of the leave products. for instance, the cis trans isomerism in alkenes can lead to different biologic activities.
Addressing these challenges requires a deep read of the molecule's construction and the reaction conditions. Chemists often use protective groups and catalysts to control the reactivity of unsaturated sites and achieve the desired outcomes.
to summarize, sites of unsaturation play a polar role in organic chemistry, influence the reactivity, constancy, and biological activity of molecules. By understand these sites and their behaviors, chemists can design and synthesise new compounds with tailor-make properties. Whether in pharmaceuticals, polymers, or natural products, the study of unsaturated sites continues to motor founding and discovery in the battlefield of chemistry.
Related Terms:
- 1 degree of unsaturation
- how to compute unsaturation
- degree of unsaturation chemistry
- 4 degrees of unsaturation representative
- degree of unsaturation definition
- zero degrees of unsaturation