Carbon, the sixth element in the periodical table, is a key building block of life and a cornerstone of mod chemistry. Understanding the valence electrons of carbon is essential for grasp its chemical behaviour and the vast array of compounds it forms. This post delves into the properties of carbon, its valence electrons, and their significance in chemic bonding and molecular structures.
Understanding Carbon and Its Valence Electrons
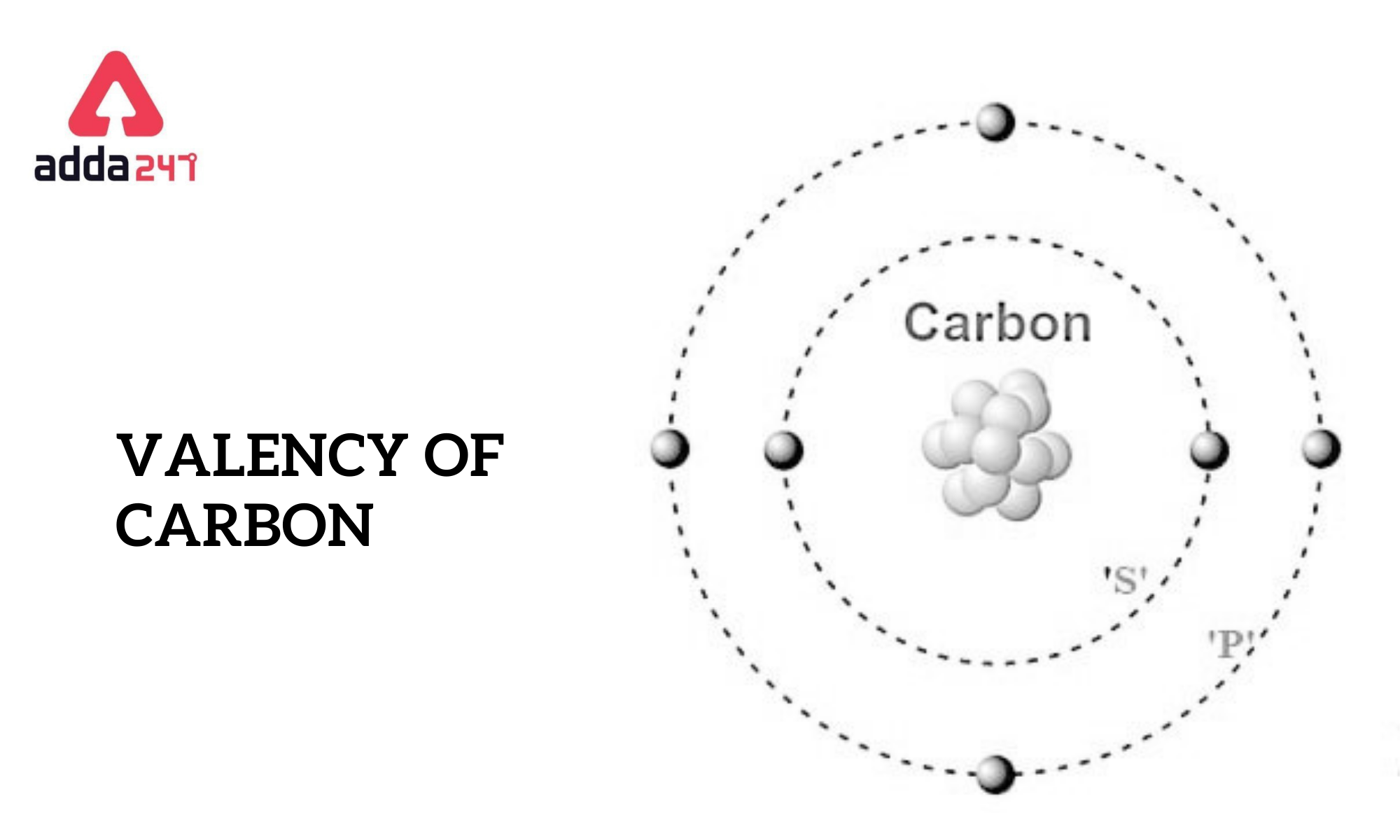
Carbon is a nonmetal with the nuclear figure 6, imply it has six protons and six electrons. The electronic form of carbon is 1s 2 2s2 2p2. The outermost shell, which contains the valency electrons, has four electrons. These valence electrons are all-important for carbon's ability to form covalent bonds with other atoms.
The Role of Valence Electrons in Chemical Bonding
The valency electrons of carbon play a pivotal role in its chemical reactivity. Carbon can form four covalent bonds, which allows it to make a all-inclusive variety of molecules. This versatility is because carbon's valency electrons can participate in single, double, and triple bonds. for instance:
- In methane (CH 4 ), carbon forms four single bonds with hydrogen atoms.
- In ethylene (C 2 H4 ), carbon forms a double bond with another carbon atom and single bonds with hydrogen atoms.
- In acetylene (C 2 H2 ), carbon forms a triple bond with another carbon atom and single bonds with hydrogen atoms.
This power to form multiple types of bonds is what makes carbon such a versatile element in organic chemistry.
Carbon's Hybridization and Molecular Geometry
Carbon's valency electrons can undergo crossing, which is the mixing of nuclear orbitals to form new hybrid orbitals. This operation is essential for understanding the geometry of carbon moderate molecules. The most mutual types of hybridization in carbon are:
- sp 3 hybridizing: This occurs when one s orbital and three p orbitals mix to form four sp 3 hybrid orbitals. This type of hybridization is seen in methane (CH 4 ), where the carbon atom forms four single bonds with hydrogen atoms, resulting in a tetrahedral geometry.
- sp 2 hybridization: This occurs when one s orbital and two p orbitals mix to form three sp 2 hybrid orbitals. This type of cross is seen in ethylene (C 2 H4 ), where the carbon atoms form a double bond with each other and single bonds with hydrogen atoms, resulting in a trigonal planar geometry.
- sp hybridization: This occurs when one s orbital and one p orbital mix to form two sp hybrid orbitals. This type of hybridization is seen in acetylene (C 2 H2 ), where the carbon atoms form a triple bond with each other and single bonds with hydrogen atoms, resulting in a linear geometry.
Understanding these cross states is all-important for omen the molecular geometry and properties of carbon bear compounds.
The Importance of Carbon in Organic Chemistry
Carbon's unique ability to form stable covalent bonds with other carbon atoms and a variety of other elements makes it the backbone of organic chemistry. Organic compounds, which are crucial for life, are primarily composed of carbon and hydrogen, with extra elements like oxygen, nitrogen, and sulfur. The valence electrons of carbon enable the formation of complex molecular structures, including:
- Hydrocarbons: Compounds composed alone of carbon and hydrogen, such as methane, ethane, and benzene.
- Alcohols: Compounds containing a hydroxyl group (OH) attach to a carbon atom, such as ethanol and methanol.
- Amines: Compounds containing an amino group (NH 2 ) attached to a carbon atom, such as methylamine and ethylamine.
- Carboxylic acids: Compounds containing a carboxyl group (COOH) attached to a carbon atom, such as acetic acid and propionic acid.
These compounds are fundamental to diverse industries, include pharmaceuticals, agriculture, and materials skill.
Carbon Allotropes and Their Properties
Carbon exists in several allotropic forms, each with unique properties ascertain by the arrangement of carbon atoms and their valency electrons. The most well known allotropes of carbon are:
- Diamond: In diamond, each carbon atom is bond to four other carbon atoms in a tetrahedral arrangement, forming a three dimensional network. This results in a very hard and stable structure.
- Graphite: In graphite, carbon atoms are arranged in hexagonal layers, with each atom tie to three others in a planar arrangement. The layers are held together by weak van der Waals forces, allowing them to slide over each other, which gives graphite its lubricating properties.
- Fullerenes: Fullerenes are molecular forms of carbon, such as buckminsterfullerene (C 60 ), which consist of closed cages of carbon atoms. These structures have unique electronic and mechanical properties.
- Graphene: Graphene is a single stratum of graphite, dwell of a hexagonal lattice of carbon atoms. It has exceptional strength, conductivity, and tractability, do it a promising material for several applications.
Each of these allotropes has distinct properties due to the different ways in which the valence electrons of carbon are utilize in bonding.
Carbon in Biological Systems
Carbon is the indispensable element in all known life forms. Its ability to form complex molecules is all-important for the structure and function of biologic systems. Key biological molecules that rely on carbon's valence electrons include:
- Proteins: These are large, complex molecules made up of amino acids, which are connect by peptide bonds formed between the valence electrons of carbon and nitrogen atoms.
- Nucleic acids: DNA and RNA are polymers of nucleotides, which contain carbon found sugars, phosphate groups, and nitrogenous bases. The valency electrons of carbon play a critical role in the formation of these molecules and their interactions.
- Lipids: These are fatty acids and other molecules that carry long hydrocarbon chains. The valency electrons of carbon enable the formation of these chains and their interactions with other molecules.
- Carbohydrates: These are sugars and starches that provide energy for living organisms. The valence electrons of carbon are involved in the shaping of glycosidic bonds between sugar molecules.
In biological systems, the valence electrons of carbon are essential for the constitution of these complex molecules and their interactions, which are vital for life processes.
Carbon's Environmental Impact
Carbon plays a significant role in the environment, peculiarly in the context of climate vary. Carbon dioxide (CO 2 ), a greenhouse gas, is a product of carbon's oxidation. The burning of fossil fuels, which are primarily composed of carbon, releases large amounts of CO2 into the atmosphere, contributing to orbicular warming. Understanding the valence electrons of carbon and their role in chemic reactions is all-important for developing strategies to extenuate carbon emissions and combat climate alter.
Additionally, carbon is a key component of organic matter in soil, which is all-important for plant growth and soil health. The cycling of carbon through ecosystems, involving processes like photosynthesis and ventilation, is key to the Earth's biosphere.
Carbon's environmental impact is a complex issue that requires a deep understanding of its chemical properties and the role of its valence electrons in respective processes.
Note: The environmental wallop of carbon is a broad and multifaceted topic that encompasses many scientific disciplines, including chemistry, biology, and environmental science.
Carbon in Industrial Applications
Carbon's unparalleled properties make it priceless in various industrial applications. Some of the key uses of carbon in industry include:
- Steel Production: Carbon is a essential component in the product of steel, where it is used to tone and indurate the metal. The valence electrons of carbon form potent bonds with iron atoms, enhancing the material's properties.
- Electronics: Carbon ground materials, such as graphene and carbon nanotubes, are used in electronics due to their olympian conductivity and mechanical strength. These materials are all-important for developing advance electronic devices and components.
- Energy Storage: Carbon is used in batteries and supercapacitors, where its valence electrons play a role in storing and unloosen electric energy. Graphite, for representative, is a key component in lithium ion batteries.
- Chemical Industry: Carbon is a fundamental element in the production of various chemicals, include plastics, pharmaceuticals, and fertilizers. The valence electrons of carbon enable the formation of complex molecular structures essential for these products.
In these industrial applications, the valency electrons of carbon are harnessed to create materials and products with specific properties and functions.
Future Directions in Carbon Research
Research on carbon continues to evolve, driven by the need to read its properties and applications wagerer. Some of the stimulate areas of research include:
- Nanotechnology: The study of carbon ground nanomaterials, such as graphene and carbon nanotubes, is a chop-chop growing field. These materials have unequaled properties that make them suitable for various applications, including electronics, energy storage, and materials skill.
- Carbon Capture and Storage: Developing technologies to seizure and store carbon dioxide from industrial processes and power plants is important for mitigating climate change. Understanding the valence electrons of carbon and their role in chemic reactions is all-important for advancing these technologies.
- Biomaterials: Carbon based materials are being explored for use in biomedical applications, such as drug delivery, tissue engineering, and aesculapian implants. The unique properties of carbon create it an ideal material for these applications.
These research areas spotlight the ongoing importance of understanding carbon's properties and the role of its valency electrons in assorted scientific and technical fields.
Carbon's versatility and the unparalleled properties of its valency electrons get it a cornerstone of modern chemistry and materials science. From its role in biologic systems to its applications in industry and engineering, carbon continues to be a subject of intense study and instauration. The valence electrons of carbon are at the heart of its chemic demeanour, enabling the shaping of complex molecules and materials with diverse properties and functions.
As our understanding of carbon deepens, so too does our ability to harness its likely for direct world-wide challenges and advance technical progress. The study of carbon and its valence electrons is a testament to the ability of scientific inquiry and the endless possibilities it offers for discovery and invention.
Related Terms:
- valence electrons of hydrogen
- valency electrons of oxygen
- valence electrons of sodium
- valency electrons of nitrogen
- valency electron of carbon atom